The demand for hygienic and odor-controlling polymer products has increased substantially across healthcare, food packaging, consumer goods, and building material applications. Antibacterial and deodorant masterbatch produced through twin screw extrusion technology provides effective antimicrobial protection and odor neutralization in polymer applications where hygiene and freshness are paramount concerns. This comprehensive guide covers formulation considerations, production processes, equipment selection, and operational best practices for manufacturing high-performance antibacterial and deodorant masterbatch.
Introduction
Microbial growth on polymer surfaces creates numerous problems including odor generation, surface discoloration, staining, and potential health hazards in sensitive applications. Antibacterial masterbatch incorporates antimicrobial agents that inhibit or eliminate bacterial and fungal growth, extending product freshness and service life while maintaining hygienic surface conditions.
Deodorant masterbatch addresses odor concerns through odor absorber incorporation and fragrance encapsulation that neutralize or mask unpleasant smells. These formulations prove essential for applications where polymer materials may be exposed to organic materials or environmental conditions favoring odor development.
The global emphasis on hygiene and wellness has driven increasing adoption of antimicrobial polymer products in medical devices, food preparation surfaces, textile fibers, and consumer products. Twin screw extrusion technology provides the precise processing conditions necessary for incorporating sensitive antimicrobial additives while maintaining their effectiveness.
Understanding Antimicrobial Mechanisms
Antimicrobial agents function through multiple mechanisms that damage or kill microorganisms on polymer surfaces. Contact killing mechanisms involve release of antimicrobial ions or compounds from the surface that destroy bacterial cell walls and membranes upon contact.
Diffusion-based mechanisms involve sustained release of antimicrobial compounds that create zones of inhibition preventing bacterial growth near the polymer surface. These systems provide residual protection as fresh antimicrobial agent diffuses to the surface over time.
Photocatalytic mechanisms utilize light-activated catalysts that generate reactive oxygen species capable of destroying microorganisms. These systems provide self-sterilizing surfaces that maintain antimicrobial activity through light exposure.
Formulation Ratio for Antibacterial and Deodorant Masterbatch
Silver-Based Antimicrobial Formulations
Silver ions provide broad-spectrum antimicrobial activity with excellent safety profiles and established efficacy against bacteria, fungi, and viruses. Silver-based masterbatch formulations offer long-lasting protection suitable for medical and food contact applications.
Typical silver-based antimicrobial masterbatch formulations contain 5 to 20 percent silver-based active ingredient, 65 to 85 percent carrier resin, and 5 to 15 percent co-additives including carriers, dispersants, and processing stabilizers. Silver zeolite and silver phosphate glass represent common commercial forms providing controlled silver ion release.
The specific silver concentration depends on required antimicrobial efficacy levels and regulatory restrictions governing silver content in specific applications. Food contact applications typically require lower silver concentrations than medical device applications.
Organic Antimicrobial Formulations
Organic antimicrobial agents including triclosan, triclocarban, and quaternary ammonium compounds provide effective antimicrobial activity at relatively low addition levels. These materials offer cost-effective protection for consumer product applications.
Typical organic antimicrobial masterbatch formulations contain 5 to 15 percent organic antimicrobial active ingredient, 70 to 85 percent carrier resin, and 5 to 10 percent processing aids ensuring adequate dispersion and stability. Triclosan and nano-zinc pyrithione represent widely used organic antimicrobial options.
Regulatory status of organic antimicrobials varies by region and application, requiring careful attention to permitted uses in target markets. Some traditional organic antimicrobials face increasing restrictions due to environmental and health concerns.
Zinc and Copper-Based Antimicrobial Formulations
Zinc and copper ions provide antimicrobial activity through multiple mechanisms including membrane disruption and enzyme inhibition. These metal-based systems offer broad-spectrum protection with favorable safety profiles and regulatory acceptance in food contact applications.
Typical zinc-based antimicrobial masterbatch formulations include 10 to 30 percent zinc-based active ingredient such as zinc pyrithione or zinc oxide nanoparticles, 55 to 75 percent carrier resin, and 5 to 15 percent dispersants and processing aids. Zinc pyrithione provides particularly effective antifungal activity alongside antibacterial performance.
Copper-based formulations typically contain 5 to 15 percent copper-based active ingredient such as copper zeolite or copper nanoparticles, with carrier resin and processing additives comprising the balance. Copper provides excellent antimicrobial efficacy with the additional advantage of integral antiviral activity.
Deodorant Masterbatch Formulations
Deodorant masterbatch incorporates odor-absorbing materials and fragrance systems that address odor concerns in polymer applications. Activated carbon, zeolites, and zinc compounds provide physical and chemical odor absorption mechanisms.
Activated carbon deodorant masterbatch formulations typically contain 15 to 30 percent activated carbon with specialized surface treatment, 60 to 75 percent carrier resin, and 5 to 10 percent dispersants ensuring uniform carbon distribution. Activated carbon provides broad-spectrum odor adsorption for demanding applications.
Fragrance-encapsulated deodorant formulations incorporate microencapsulated fragrances that release scent upon friction or degradation. These systems provide ongoing freshness perception rather than odor absorption, addressing applications where pleasant scent provides consumer benefit.
Combination Antimicrobial and Deodorant Systems
Advanced formulations combine antimicrobial and deodorant functionality for comprehensive odor control. These synergistic systems address both the source of odors through antimicrobial action and any residual odors through chemical or physical absorption.
Typical combination formulations include 8 to 15 percent antimicrobial active ingredient, 10 to 20 percent deodorant component such as activated carbon or zinc compounds, with carrier resin and processing additives comprising the balance. The specific ratio depends on application requirements and regulatory constraints.
Production Process for Antibacterial and Deodorant Masterbatch
Raw Material Handling and Preparation
Antimicrobial masterbatch production requires careful attention to raw material handling that protects both product quality and worker safety. Many antimicrobial additives are supplied as powders requiring dust control measures during feeding and transfer operations.
Carrier resin preparation addresses moisture content and physical form requirements for optimal extrusion processing. Resin drying ensures moisture levels below specification limits for the specific polymer type, preventing hydrolysis during processing that could compromise mechanical properties.
Additive powder handling requires attention to accurate dosing and uniform distribution. Closed transfer systems minimize airborne dust while ensuring complete transfer of antimicrobial additives to mixing equipment. Gravimetric feeding systems provide precise additive introduction rates that maintain formulation accuracy.
Pre-Mixing Operations
Pre-mixing ensures uniform antimicrobial distribution before extrusion processing. The pre-mixing stage addresses the challenge of incorporating concentrated additive formulations into a homogeneous carrier matrix.
High-intensity mixing equipment provides rapid blending of antimicrobial concentrates with carrier resin. Mixing time optimization balances thorough distribution against potential damage from excessive shear forces. Visual inspection of pre-mix uniformity provides quality verification before extrusion processing.
Masterbatch concentrates may be produced at higher antimicrobial concentrations during pre-mixing, with subsequent let-down into appropriate carrier systems for specific applications. This approach enables manufacturing flexibility while maintaining consistent additive quality.
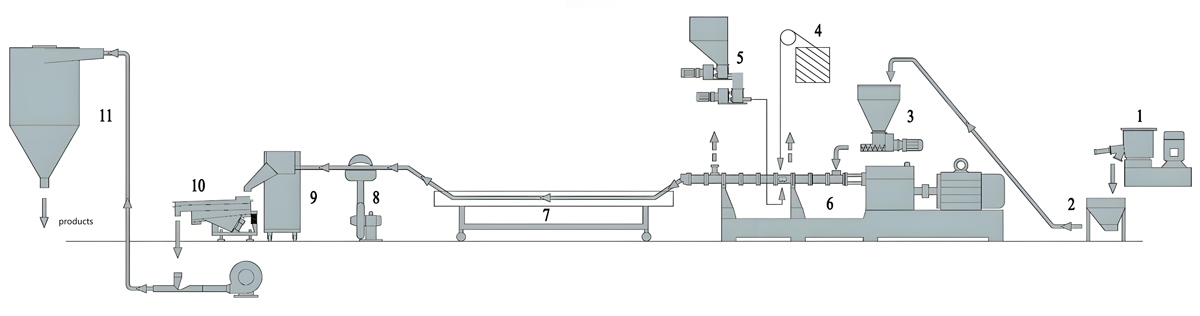
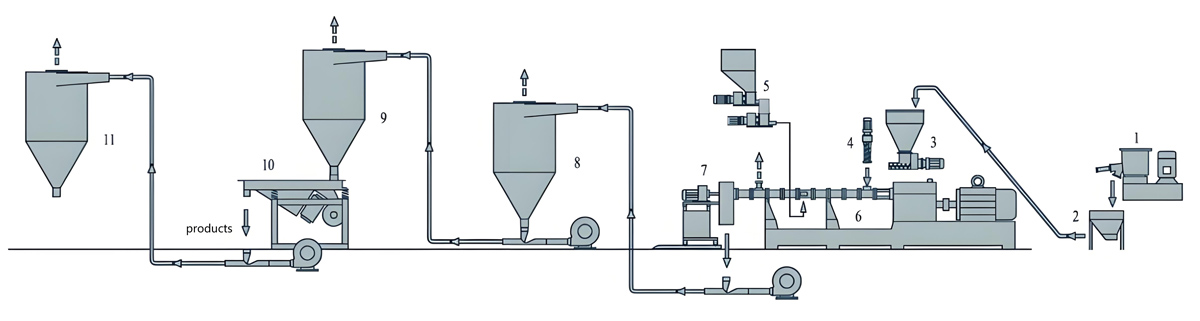
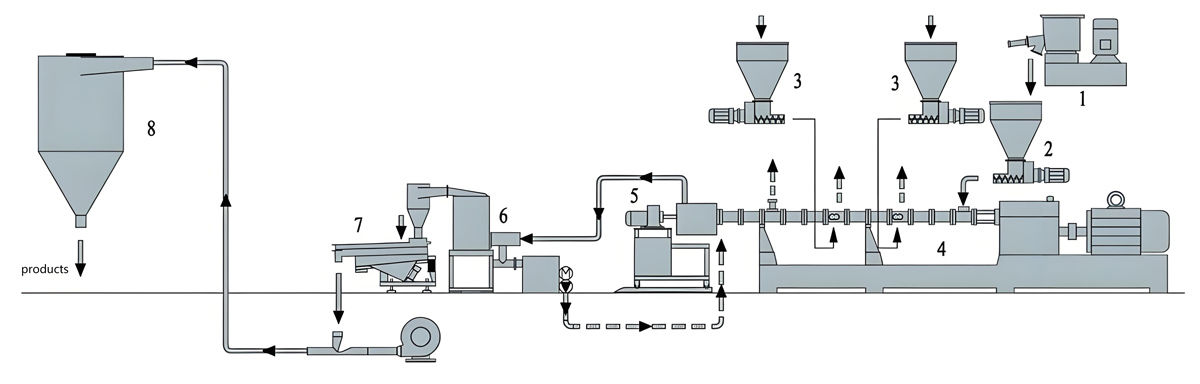
Twin Screw Extrusion Processing
The twin screw extrusion process transforms pre-mixed formulations into homogeneous masterbatch pellets suitable for downstream application. Process optimization addresses additive dispersion, thermal stability of sensitive antimicrobials, and production efficiency requirements.
Feeding zones introduce pre-mixed materials at controlled rates that maintain appropriate extruder fill levels. Gravimetric feeding systems provide continuous formulation verification through real-time weight monitoring of introduced materials. Multiple feed points enable sequential addition of heat-sensitive antimicrobials at barrel locations offering optimal thermal exposure.
Mixing zones incorporate specialized screw elements that distribute antimicrobial additives throughout the polymer matrix while minimizing thermal stress on sensitive components. Kneading blocks and mixing elements create flow patterns necessary for thorough additive dispersion without degradation from excessive shear heating.
The pumping zone delivers consistent melt flow to the die plate while maintaining temperature conditions that preserve antimicrobial effectiveness. Pressure development in this zone forces melt through the die plate openings, creating pellets through underwater or water ring pelletizing systems.
Pelletizing and Post-Processing
Pelletizing systems transform extruded melt into uniform pellet forms suitable for handling, storage, and subsequent application. Underwater pelletizing provides the most consistent pellet quality for antimicrobial masterbatch production.
Pellet cooling must be controlled to prevent thermal shock that could damage surface treatments or encapsulated fragrance systems. Water temperature and flow control provide gentle cooling that maintains pellet integrity and antimicrobial distribution.
Packaging operations protect finished masterbatch from environmental exposure that could compromise antimicrobial effectiveness. Airtight containers prevent moisture uptake and preserve additive stability during storage and transportation.
Production Equipment Introduction
Kerke KTE Series Twin Screw Extruders
Kerke KTE series twin screw extruders provide the processing capabilities required for high quality antibacterial and deodorant masterbatch production. The series encompasses models suited to various production scales from laboratory development through industrial volume manufacturing.
The KTE-36B extruder features 35.6mm screw diameter and achieves throughput rates of 20 to 100kg per hour. This compact machine suits formulation development, pilot production, and small volume specialty masterbatch manufacturing. The precise control capabilities enable optimization of sensitive antimicrobial formulations.
The KTE-50B extruder provides 50.5mm screw diameter with production rates of 80 to 200kg per hour. This mid-range model balances processing quality with production efficiency for moderate volume commercial production.
The KTE-65B extruder offers 62.4mm screw diameter and throughput of 200 to 450kg per hour. This capacity level addresses commercial production requirements while maintaining the mixing efficiency necessary for uniform additive dispersion.
The KTE-75B extruder features 71mm screw diameter with production capabilities of 300 to 800kg per hour. This higher capacity model suits established masterbatch suppliers serving volume hygiene product markets.
The KTE-95D extruder provides the largest production capacity in the series with 93mm screw diameter and throughput of 1000 to 2000kg per hour. This industrial scale machine meets high volume production requirements for major hygiene and packaging polymer markets while maintaining quality consistency.
Feeding and Handling Systems
Gravimetric feeding systems provide the precise ingredient introduction rates required for consistent antimicrobial masterbatch formulations. These systems incorporate loss-in-weight technology that continuously measures and adjusts feed rates to maintain formulation accuracy.
Additive feeding equipment handles both liquid and powder antimicrobials with appropriate delivery mechanisms. Powder feeders require careful design to prevent segregation and ensure uniform additive introduction throughout production runs.
Material handling systems transfer raw materials from storage to production equipment and finished product to packaging. Closed transfer systems protect sensitive antimicrobials from environmental exposure during handling operations.
Pelletizing and Packaging Equipment
Underwater pelletizing systems produce the spherical pellets preferred for antimicrobial masterbatch applications. These systems maintain precise water temperature and flow control to produce pellets with consistent geometry and surface quality.
Centrifugal dryers remove surface water while minimizing mechanical stress that could damage pellets. Integrated screens classify pellets by size and remove fines that could cause feeding problems in downstream applications.
Metal detection and quality verification systems provide inspection that identifies contaminated or defective product. These systems protect product quality and customer satisfaction in demanding hygiene applications.
Parameter Settings for Antibacterial and Deodorant Masterbatch
Temperature Profile Optimization
Temperature profile configuration directly impacts masterbatch quality by affecting melt viscosity, additive stability, and dispersion efficiency. Profile optimization balances processing requirements against thermal stability limitations of sensitive antimicrobial additives.
Typical temperature profiles for silver-based antimicrobial masterbatch production begin at 180 to 200 degrees Celsius in the feeding zone, increase to 200 to 220 degrees through primary mixing sections, and maintain 200 to 210 degrees in pumping and die zones. The specific profile requires adjustment based on carrier resin, additive package sensitivity, and throughput rate.
Organic antimicrobial formulations may require lower processing temperatures to prevent thermal degradation. Triclosan and similar compounds show sensitivity to elevated temperatures, requiring profile adjustment to preserve antimicrobial activity.
Deodorant formulations containing activated carbon or zeolites tolerate higher processing temperatures since these mineral components exhibit excellent thermal stability. Carrier resin thermal stability limits processing temperatures more than deodorant additive constraints.
Screw Speed and Shear Control
Screw speed selection influences mixing efficiency, melt temperature development, and additive integrity. Higher speeds increase shear rates that improve dispersion but also increase thermal stress on sensitive antimicrobials.
Optimal screw speeds for antimicrobial masterbatch typically range from 150 to 350 revolutions per minute depending on formulation, equipment configuration, and quality requirements. Lower speeds protect heat-sensitive additives while higher speeds enable faster production rates when additive stability permits.
Screw element selection creates specific mixing intensity profiles throughout the barrel. Kneading blocks generate high shear zones for breaking up additive agglomerates. Conveying elements transport materials through the barrel while mixing elements provide the dispersion required for uniform antimicrobial distribution.
Residence Time and Throughput Balance
Residence time within the extruder must provide sufficient thermal and mechanical energy for complete melting and additive dispersion while avoiding degradation of sensitive antimicrobials. Mean residence times of 30 to 90 seconds typically achieve adequate processing without excessive thermal exposure.
Throughput rate selection must consider the extruder fill level required for effective mixing. Insufficient fill creates poor mixing quality while excessive fill causes pressure instability and potential quality variation. Throughput and screw speed coordination maintains optimal fill levels across production rate requirements.
Narrow residence time distribution improves product consistency by ensuring all material receives similar processing. Screw design optimization through barrier elements and specialized pumping sections narrows the distribution and reduces product quality variation.
Vacuum and Ventilation Settings
Vacuum ventilation removes moisture and volatile degradation products from the melt. Vacuum levels of 50 to 100 millibars effectively remove volatiles without excessive polymer loss through the vent system.
Vent zone placement requires consideration of when volatiles reach maximum concentration during melting. Positioning vents downstream of melting zones captures moisture released as polymers transition from solid to melt state.
Excessive vacuum can draw fine additive particles from the melt, creating formulation variation and potentially damaging vacuum equipment. Vacuum level optimization balances volatile removal efficiency against material loss and additive retention requirements.
Equipment Price
Equipment investment for antimicrobial and deodorant masterbatch production varies based on production capacity requirements and automation levels. The Kerke KTE series provides equipment options spanning research to industrial production scales.
The KTE-36B twin screw extruder represents the entry level investment at $25,000 to $35,000. This model suits research and development applications and pilot production before scale-up to commercial volumes.
The KTE-50B twin screw extruder requires an investment of $40,000 to $60,000. This mid-range equipment balances processing capability with production efficiency for moderate volume commercial production.
The KTE-65B twin screw extruder costs between $50,000 and $80,000. This model addresses commercial production requirements for established antimicrobial masterbatch suppliers.
The KTE-75B twin screw extruder commands prices ranging from $70,000 to $100,000. This higher capacity model suits dedicated production facilities serving volume hygiene product markets.
The KTE-95D twin screw extruder represents the premium equipment tier with prices of $120,000 to $200,000. This industrial scale machine enables high volume production for major hygiene and packaging polymer markets while maintaining quality consistency.
Complete production system investment extends beyond extruder purchase to include feeding systems, pelletizing equipment, material handling, quality control instrumentation, and facility requirements. Total system investments typically range from 1.5 to 2.5 times the extruder purchase price depending on automation level and quality system requirements.
Problems in Production Process and Solutions
Antimicrobial Activity Loss During Processing
Problem Description: Thermal or shear degradation of antimicrobial additives during extrusion reduces their effectiveness in finished applications. This manifests as inadequate antimicrobial performance in certified products and potential regulatory compliance issues.
Root Cause Analysis: Excessive barrel temperatures, extended residence time at elevated temperatures, or excessive shear heating can degrade sensitive antimicrobial compounds. Some organic antimicrobials particularly may decompose when exposed to excessive thermal stress.
Solution Approach: Reduce barrel temperatures throughout the profile, particularly in zones containing antimicrobial additives. Increase throughput rate to reduce material residence time in high temperature zones. Modify screw configuration to reduce shear heating in sensitive zones. Consider split feeding where heat-sensitive antimicrobials are introduced downstream of the highest temperature zones.
Prevention Methods: Establish validated temperature profiles and throughput limits for each antimicrobial formulation based on additive thermal stability data. Implement temperature monitoring with alarms that identify excursions before significant degradation occurs. Document process parameters for each production run to enable traceability and optimization.
Incomplete Additive Dispersion
Problem Description: Poor dispersion creates localized areas of excessive antimicrobial concentration adjacent to areas with inadequate protection. This inconsistency manifests as variable antimicrobial protection performance and potential quality failures in testing.
Root Cause Analysis: Insufficient mixing intensity, inadequate pre-mixing, or formulation issues causing additive agglomeration can all produce dispersion problems. High viscosity formulations with elevated antimicrobial loadings particularly challenge dispersion efficiency.
Solution Approach: Review screw element configuration to ensure adequate mixing elements in zones where antimicrobials are present. Verify pre-mixing equipment operation and mixing times. Consider higher intensity mixing configurations for difficult-to-disperse additives. Implement pre-blending procedures that ensure uniform additive distribution before extrusion.
Prevention Methods: Establish standard screw configurations for each formulation type based on accumulated experience. Monitor product quality trends to identify gradual degradation indicating equipment wear. Conduct periodic dispersion efficiency validation testing using standardized procedures.
Fragrance Loss in Deodorant Masterbatch
Problem Description: Volatilization of encapsulated or adsorbed fragrances during extrusion processing reduces sensory performance of deodorant masterbatch. This results in products with minimal freshness perception despite adequate deodorant component loading.
Root Cause Analysis: Excessive processing temperatures cause volatilization of fragrance compounds before encapsulation can protect them. Shear stress during mixing can rupture microcapsules before extrusion is complete. Extended residence time at elevated temperatures increases fragrance losses.
Solution Approach: Reduce processing temperatures throughout the barrel profile to minimize fragrance volatilization. Modify screw configuration to minimize shear stress on encapsulated fragrances. Consider downstream fragrance addition where possible to avoid processing losses entirely.
Prevention Methods: Select fragrance compounds with higher thermal stability for extrusion processing. Verify encapsulation quality before use in formulations. Establish processing parameters validated for specific fragrance types and concentrations.
Color Variation and Discoloration
Problem Description: Color variation between production batches or within individual batches creates quality concerns for applications where appearance specifications may apply. This also indicates potential formulation inconsistency that could affect antimicrobial performance.
Root Cause Analysis: Additive impurities, cross-contamination from previous productions, or degradation reactions can produce color shifts. Some antimicrobial additives, particularly silver-based systems, may exhibit color bodies that create visible effects in finished masterbatch.
Solution Approach: Implement comprehensive cleaning procedures between production runs. Verify raw material quality through incoming inspection testing. Adjust processing conditions to minimize thermal stress on color-sensitive additives. Consider alternative additive grades with improved color stability.
Prevention Methods: Establish cleaning validation protocols specifying procedures for different formulation transitions. Maintain raw material specifications and incoming quality verification. Document color measurements for each production lot to enable trend analysis and rapid problem identification.
Regulatory Compliance Issues
Problem Description: Antimicrobial masterbatch intended for food contact or medical applications may fail regulatory compliance testing due to formulation or processing issues. These failures create significant customer and regulatory concerns.
Root Cause Analysis: Incorrect additive selection, migration exceeding regulatory limits, or processing conditions affecting additive form can all create compliance issues. Formulation errors, cross-contamination, or documentation failures may also result in compliance failures.
Solution Approach: Verify that all formulation components are approved for intended applications and regulatory jurisdictions. Conduct migration testing on production samples to verify compliance before commercial release. Review documentation and traceability systems to ensure complete compliance documentation.
Prevention Methods: Maintain approved formulations for each target regulatory jurisdiction. Implement quality systems that verify formulation accuracy and traceability. Conduct regular compliance testing on production samples to verify continued compliance.
Maintenance
Daily Operational Maintenance
Daily maintenance activities ensure consistent product quality and equipment reliability for antimicrobial masterbatch production. Operator inspections at shift start verify equipment readiness and identify any issues requiring attention before production begins.
Temperature controller verification confirms accurate temperature measurement and control throughout the barrel profile. Recording actual temperatures against setpoints enables trend analysis that identifies developing problems. Any significant deviations require investigation and correction before production continues.
Feeding system inspection verifies smooth material flow and proper function of feeding equipment. Hopper level monitoring ensures adequate material supply throughout production runs. Closed systems inspection confirms that handling integrity is maintained throughout production.
Die plate inspection and cleaning removes accumulated material that could restrict flow or cause contamination. Visual inspection of startup pellets verifies product quality before releasing production for packaging.
Weekly Maintenance Activities
Weekly maintenance extends equipment care beyond daily activities to address components requiring less frequent attention. Gravimetric feeding system calibration verification using certified test weights confirms continued accuracy within specification limits.
Vacuum system inspection examines filters, seals, and connections for condition and proper function. Filter inspection and replacement maintains vacuum efficiency and prevents material loss through the vent system.
Screw torque indicator inspection identifies any loosening that could allow element rotation during operation. Torque measurements provide baseline data for detecting changes over time indicating potential mechanical issues.
Pelletizing system inspection examines knife wear, die plate condition, and water system function. Regular knife sharpening maintains cutting quality while die inspection identifies wear patterns requiring attention.
Monthly and Quarterly Maintenance
Monthly maintenance addresses wear patterns and component conditions requiring periodic assessment. Barrel and screw wear inspection identifies sections approaching replacement thresholds based on accumulated operating hours and historical wear rates.
Gearbox oil analysis provides early warning of contamination or degradation that could lead to mechanical failure. Oil replacement schedules maintain transmission health while analysis results guide maintenance timing decisions.
Electrical system inspection examines connections, motor condition, and control system calibration. Temperature controller calibration verification ensures accurate temperature measurement throughout the barrel profile.
Quarterly maintenance encompasses comprehensive equipment assessment addressing all major systems. Extruder barrel inspection using borescope equipment documents internal wear patterns and accumulated deposits affecting product quality. Performance qualification testing verifies equipment capability to produce product meeting specifications.
Annual and Long-Term Maintenance
Annual maintenance represents comprehensive equipment overhaul ensuring continued reliability and quality performance. Complete extruder disassembly and inspection examines all internal components for wear, damage, or accumulated material affecting performance.
Screw element replacement planning considers accumulated operating hours and historical wear patterns to optimize replacement timing. Proactive replacement prevents quality problems emerging from excessive wear while enabling scheduled maintenance during planned downtime.
Control system updates and calibration ensure continued accurate equipment operation. Process parameter documentation enables rapid recovery following control system maintenance or upgrades.
Performance qualification testing documents equipment capability for quality system compliance purposes. These records demonstrate continued ability to produce antimicrobial masterbatch meeting demanding hygiene requirements for food contact and medical applications.
FAQ
What antimicrobial agents are most effective for polymer applications?
Silver-based antimicrobials provide excellent broad-spectrum activity with good regulatory acceptance across most applications. Zinc pyrithione offers effective antifungal performance suitable for outdoor and humid environment applications. Triclosan provides cost-effective antibacterial activity though regulatory restrictions apply in some regions. Silver zeolite, silver phosphate glass, and nano-zinc oxide represent commonly used commercial forms providing controlled antimicrobial ion release.
How do antimicrobial masterbatch loadings affect polymer properties?
High antimicrobial loadings can affect polymer mechanical properties depending on additive type and loading level. Silver-based systems typically have minimal impact on polymer properties at normal usage levels. Zinc and copper systems may affect color and certain mechanical properties at higher concentrations. Organic antimicrobials generally have minimal impact on polymer properties at effective addition levels.
What carrier resins work best for antimicrobial masterbatch?
Carrier resin selection depends on final application polymer compatibility and processing requirements. Polypropylene and polyethylene carriers offer broad compatibility and cost-effective performance. EVA carriers provide excellent compatibility with flexible film applications. Specialty carriers including polyamide and polyester address specific polymer application requirements.
How long do antimicrobial effects last in finished products?
Antimicrobial effectiveness duration depends on additive type, concentration, and application conditions. Silver-based systems provide sustained antimicrobial activity through controlled silver ion release over extended periods. Surface rubbing and cleaning can remove surface antimicrobials, reducing effectiveness over time. Migration-based systems may provide longer residual activity as fresh antimicrobial agent migrates to the surface.
What quality control tests are essential for antimicrobial masterbatch?
Essential quality tests include antimicrobial content verification through analytical methods, minimum inhibitory concentration testing against target organisms, moisture content determination, and dispersion evaluation. Particle size distribution analysis verifies pellet characteristics. Regulatory compliance testing confirms suitability for intended food contact or medical applications.
Can I combine multiple antimicrobial agents in one formulation?
Synergistic combinations of multiple antimicrobial agents can enhance overall effectiveness while potentially reducing required addition levels. Common synergistic combinations include silver with zinc compounds and organic antimicrobials with metal-based systems. Compatibility testing verifies that combined antimicrobials do not interact negatively during processing or in final applications.
How should I store antimicrobial masterbatch?
Antimicrobial masterbatch storage requires protection from moisture, heat, and direct light that could degrade additive effectiveness over extended periods. Airtight packaging prevents moisture uptake and preserves additive stability. FIFO inventory practices ensure oldest material is used first. Storage at moderate temperatures away from direct sunlight maintains product quality during storage periods of 12 to 24 months.
What regulatory approvals apply to antimicrobial masterbatch?
Regulatory requirements vary by application and target market. Food contact applications require compliance with FDA regulations in the United States and similar authorities in other regions. Medical device applications require biocompatibility testing and device-specific regulatory approval. EPA registration may be required for antimicrobial claims in certain applications. European Biocidal Products Regulation affects antimicrobial products in European markets.
Conclusion
Antibacterial and deodorant masterbatch production through twin screw extrusion technology addresses growing demand for hygienic polymer products across healthcare, food packaging, consumer goods, and building material applications. The combination of effective antimicrobial agents and deodorant systems provides comprehensive protection against microbial growth and odor development that extends product freshness and service life.
Successful antimicrobial masterbatch manufacturing requires careful attention to formulation design, process optimization, and quality control. The Kerke KTE series provides processing equipment capable of producing consistent, high-quality masterbatch across the production capacity range required by different market segments.
Equipment investment considerations must account for the specialized requirements of antimicrobial production including process control precision, material handling requirements, and quality system capabilities. Supporting systems including feeding equipment, pelletizing systems, and quality control instrumentation all contribute to effective production operations.
Ongoing process optimization based on product quality feedback and maintenance activities ensure continued production of masterbatch meeting demanding hygiene requirements. Investment in operator training, quality systems, and equipment maintenance yields returns through consistent product quality and reduced production problems.
The technical knowledge presented in this guide provides a foundation for developing antimicrobial masterbatch manufacturing capabilities. Organizations pursuing opportunities in healthcare, food packaging, and consumer product applications requiring hygiene protection can leverage this knowledge to establish effective production operations serving markets demanding long-term antimicrobial performance and regulatory compliance.