Daily chemical packaging masterbatch serves the cosmetics, personal care, and household chemical industries with specialized formulations that address the unique requirements of packaging for these demanding applications. The production of masterbatch for daily chemical packaging combines aesthetic requirements for brand differentiation with functional properties essential for product protection and regulatory compliance. Twin screw extrusion technology has established itself as the preferred manufacturing method for daily chemical packaging masterbatch, providing the quality consistency, formulation flexibility, and production efficiency that this specialized market segment requires. This comprehensive guide explores the technical considerations, formulation strategies, regulatory requirements, and production practices essential for successful daily chemical packaging masterbatch manufacturing.
Introduction to Daily Chemical Packaging Masterbatch
The daily chemical industry encompasses a diverse range of products including cosmetics, skincare preparations, hair care products, fragrances, personal hygiene items, and household cleaning products. Each product category presents specific requirements for packaging materials that affect masterbatch formulation design and production specifications. The packaging serves multiple essential functions including product protection, brand communication, consumer convenience, and regulatory compliance.
Visual appearance represents a critical factor in daily chemical packaging success, as consumers make rapid purchasing decisions based on packaging aesthetics and brand perception. Masterbatch formulations must deliver consistent, vibrant colors that maintain appearance throughout product shelf life while differentiating products in competitive retail environments. Color accuracy and batch-to-batch consistency directly impact brand integrity and consumer trust.
Functional requirements for daily chemical packaging masterbatch extend beyond aesthetic considerations to include chemical resistance, barrier properties, and compatibility with packaged contents. Many daily chemical products contain ingredients that may interact with packaging materials, requiring masterbatch formulations that provide appropriate resistance while maintaining product integrity. Regulatory compliance for food contact and cosmetic packaging applications imposes additional requirements that influence formulation design and manufacturing practices.
The combination of aesthetic demands, functional requirements, and regulatory compliance creates a complex set of considerations that masterbatch producers must address through careful formulation design, validated manufacturing processes, and comprehensive quality assurance programs. Success in this market segment requires technical expertise across multiple disciplines combined with manufacturing capabilities that ensure consistent quality and regulatory compliance.
Regulatory Framework for Daily Chemical Packaging
Daily chemical packaging masterbatch production operates within a comprehensive regulatory framework that governs material safety, content compatibility, and regulatory compliance for packaged products. Understanding these requirements is essential for any producer serving the daily chemical packaging market.
Food Contact Regulations
Food contact regulations establish requirements for materials that may transfer substances to food products, including packaging materials, containers, and processing equipment. Masterbatch formulations intended for food contact applications must comply with applicable regulations in target markets, including FDA regulations in the United States and EU Regulation 10/2011 in Europe.
FDA regulations for food contact materials, codified in 21 CFR parts 175-179, establish specific requirements for polymers, colorants, and additives used in food contact applications. Colorants must be listed in the regulation or have effective food contact notifications that authorize their use. Masterbatch producers must ensure that formulations comply with applicable substance restrictions and migration limits.
EU Regulation 10/2011 establishes requirements for plastic materials intended for food contact, including positive lists of authorized substances, migration limits, and testing requirements. Compliance verification requires testing under standardized conditions that simulate intended food contact applications. The regulation establishes overall migration limits of 60 mg/kg food and specific migration limits for substances with established restrictions.
Cosmetic Packaging Regulations
Cosmetic packaging regulations vary by jurisdiction but generally establish requirements for material safety and ingredient disclosure that affect packaging material selection. In the European Union, cosmetic products are regulated under Regulation (EC) No 1223/2009, which establishes requirements for product safety without specifically regulating packaging materials. However, packaging must not compromise product safety as defined by the regulation.
The EU REACH regulation affects cosmetic packaging materials through restrictions on substances that may be present in articles, including packaging components. Masterbatch formulations must ensure compliance with applicable REACH restrictions, particularly for substances that may migrate from packaging to cosmetic products.
US cosmetic regulations, overseen by the FDA, establish requirements for cosmetic labeling and product safety without comprehensive packaging material regulations. However, cosmetic manufacturers typically impose material requirements on suppliers, including masterbatch producers, to ensure product safety and regulatory compliance.
Regional Requirements
Regional regulatory variations require attention from masterbatch producers serving multiple markets. Each major market region establishes specific requirements that may differ in substance authorizations, testing protocols, and documentation requirements.
Asian markets, including China, Japan, and South Korea, maintain specific regulations for food contact and cosmetic packaging materials. Chinese regulations GB 4806.1-2016 and associated standards establish requirements for food contact materials including comprehensive positive lists and testing requirements. Japanese and Korean regulations similarly establish specific requirements that affect formulation compliance strategies.
Documentation requirements vary by market, with some regions requiring extensive testing and certification while others rely on manufacturer declarations of compliance. Understanding specific market requirements enables efficient compliance programs that address all applicable regulations without unnecessary testing or documentation.
Formulation Ratio for Daily Chemical Packaging Masterbatch
Daily chemical packaging masterbatch formulations require careful optimization to address aesthetic requirements, functional performance, and regulatory compliance simultaneously. The following sections detail typical formulation approaches for key application categories.
Cosmetic Container Masterbatch Formulation
Cosmetic containers, including bottles, jars, and tubes for skincare, makeup, and personal care products, require masterbatch formulations that deliver premium visual appearance while maintaining chemical resistance for the packaged contents. These applications often demand high aesthetic standards that differentiate premium products in competitive retail environments.
Base resin selection for cosmetic containers typically focuses on materials with excellent clarity and surface quality, including polypropylene, polyethylene, and specialized polymer grades designed for cosmetic applications. Material selection considers chemical compatibility with specific cosmetic formulations, including potential interactions with active ingredients, fragrances, or emollients that may be present.
Pigment selection for cosmetic containers emphasizes color brilliance, light fastness, and chemical resistance. Organic pigments provide vibrant color options for premium cosmetics, while inorganic pigments offer excellent light fastness and opacity for specific color requirements. Pigment loadings typically range from 5% to 30% depending on color strength requirements and specific pigment characteristics. High-chroma pigments and specialty colorants may be incorporated at lower loadings to achieve desired color intensity.
Additive packages for cosmetic container masterbatch address specific functional requirements including UV stabilization for products with light exposure, fragrance compatibility additives, and processing aids for high-speed injection molding or extrusion. The additive package must maintain compatibility with cosmetic formulations while providing required functional properties.
Fragrance Bottle Masterbatch Formulation
Fragrance bottles present unique challenges due to the aggressive nature of fragrance formulations, which typically contain high concentrations of alcohol and aromatic compounds that may interact with packaging materials. Masterbatch formulations for fragrance bottles must provide exceptional chemical resistance alongside premium aesthetic quality.
Base resin selection for fragrance bottles emphasizes chemical resistance, with glass-recycled polyester and specialty polymer grades providing appropriate barrier properties and chemical resistance. Polypropylene and polyethylene grades with enhanced chemical resistance may also serve specific fragrance applications.
Pigment systems for fragrance bottles must demonstrate exceptional chemical resistance to prevent color changes or migration during product storage. Specialty high-fastness pigments, including certain quinacridone, perylene, and phthalocyanine pigments, provide color options with demonstrated fragrance compatibility. Low pigment loadings, typically 2% to 15%, minimize potential interaction risks while achieving desired color development.
Barrier additives may be incorporated to enhance chemical resistance and prevent fragrance component migration through packaging walls. These specialized additives reduce permeability while maintaining aesthetic quality and processing characteristics.
Household Chemical Container Masterbatch
Household chemical containers, including bottles for cleaning products, detergents, and bleach formulations, require masterbatch formulations that withstand harsh chemical environments while maintaining acceptable appearance and processing characteristics. These applications often involve aggressive chemical formulations at varying pH levels and temperature conditions.
Base resin selection for household chemical containers focuses on chemical resistance to the specific formulations being packaged. Polyethylene provides excellent resistance to many aqueous formulations, while polypropylene offers enhanced resistance to certain organic solvents and elevated temperature exposure. Specialized polymer grades address specific household chemical requirements.
Pigment systems for household chemical containers must demonstrate excellent chemical resistance to prevent color degradation during storage. Inorganic pigments typically provide superior chemical resistance compared to organic pigments, making them preferred choices for aggressive formulations. Pigment loadings range from 10% to 40% depending on color requirements and specific formulation characteristics.
Stabilizer packages for household chemical containers address UV exposure during storage and use, with carbon black and UV absorber combinations providing enhanced protection. Impact modification ensures adequate toughness for packaging durability during handling and consumer use.
Closure and Dispenser Masterbatch Formulation
Closures and dispensers for daily chemical packaging require specialized masterbatch formulations that provide functional properties essential for sealing, dispensing, and product protection. These components often involve thin sections with demanding processing requirements alongside functional performance specifications.
Base resin selection for closures typically involves polypropylene homopolymer grades with high crystallinity that provide dimensional stability and seal performance. The masterbatch carrier system must ensure compatibility with the base resin and maintain seal characteristics.
Pigment systems for closures must provide consistent color matching while maintaining functional performance requirements. Color accuracy is critical for brand identification and product differentiation, requiring precise formulation control and quality verification.
Functional additive packages for closures may include slip modifiers, anti-static agents, and sealing lubricant carriers. These additives must function effectively while maintaining regulatory compliance for food contact or cosmetic applications.
Production Process for Daily Chemical Packaging Masterbatch
Daily chemical packaging masterbatch production requires manufacturing practices that ensure quality consistency, regulatory compliance, and aesthetic performance. These requirements influence process design, equipment selection, and quality systems.
Raw Material Qualification
Raw material qualification for daily chemical packaging masterbatch ensures that all components meet applicable regulatory requirements and quality specifications. Supplier qualification programs establish approved vendor lists and incoming inspection requirements that maintain material consistency.
Colorant qualification requires comprehensive testing to verify regulatory compliance, including food contact authorization where applicable, heavy metal content verification, and migration testing under relevant conditions. Each colorant must have documented compliance status for all target markets.
Resin and additive qualification similarly requires compliance documentation, including food contact authorizations, REACH compliance statements, and safety data sheets with comprehensive hazard information. Material specifications establish acceptance criteria that ensure consistent quality across supplier lots.
Formulation Development and Color Matching
Formulation development for daily chemical packaging masterbatch combines color matching with regulatory compliance verification and performance optimization. Color matching utilizes spectrophotometric analysis to achieve precise color targets while establishing tolerance windows that guide production quality control.
Color development proceeds through sample evaluation stages, with initial laboratory batches evaluated for color accuracy, dispersion quality, and processing characteristics. Production-scale trials verify that laboratory formulations translate successfully to production conditions before full commercial production.
Regulatory compliance verification occurs throughout formulation development, ensuring that selected components have appropriate authorizations for target market applications. Compliance documentation compilation supports customer regulatory compliance requirements and regulatory inspections.
Twin Screw Compounding Process
Twin screw compounding for daily chemical packaging masterbatch follows established principles with specific attention to quality consistency and regulatory compliance requirements. Equipment configuration and process parameters ensure complete dispersion and uniform distribution of all components.
Screw configuration design addresses specific formulation requirements, with mixing sections optimized for high-pigment-load formulations and complex additive packages. Standard configurations for daily chemical packaging masterbatch typically employ L/D ratios of 40:1 to 48:1 with moderate compression ratios of 2.5:1 to 3:1.
Temperature profile optimization balances processing efficiency with thermal stability requirements. Typical processing temperatures for polypropylene-based daily chemical packaging masterbatch range from 200 to 260 degrees Celsius, with specific profiles adjusted according to formulation characteristics.
Quality Verification and Release
Quality verification for daily chemical packaging masterbatch encompasses comprehensive testing that verifies aesthetic performance, processing characteristics, and regulatory compliance. Batch release procedures ensure that only conforming product proceeds to shipment.
Aesthetic quality verification includes color measurement against reference standards, dispersion evaluation through microscopy or melt index testing, and visual inspection for contamination or defects. Statistical process control techniques monitor quality consistency throughout production.
Functional testing verifies processing characteristics through melt flow testing, density measurement, and application-specific testing as required. Regulatory compliance verification may include migration testing for food contact applications or chemical compatibility assessment for cosmetic packaging.
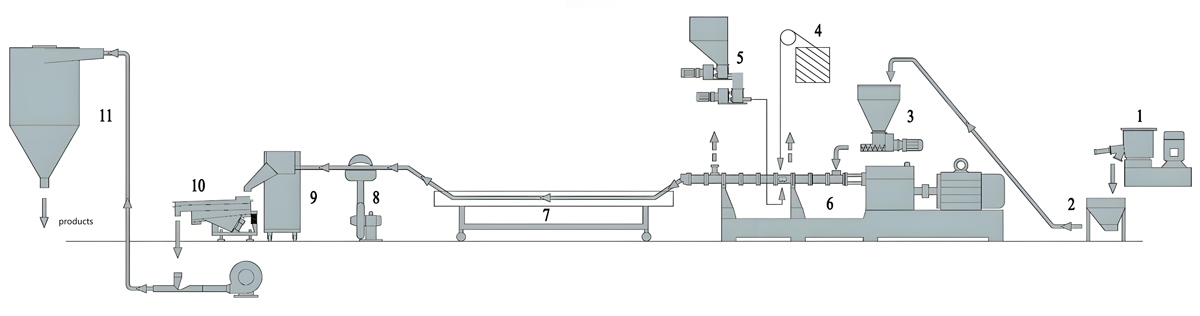
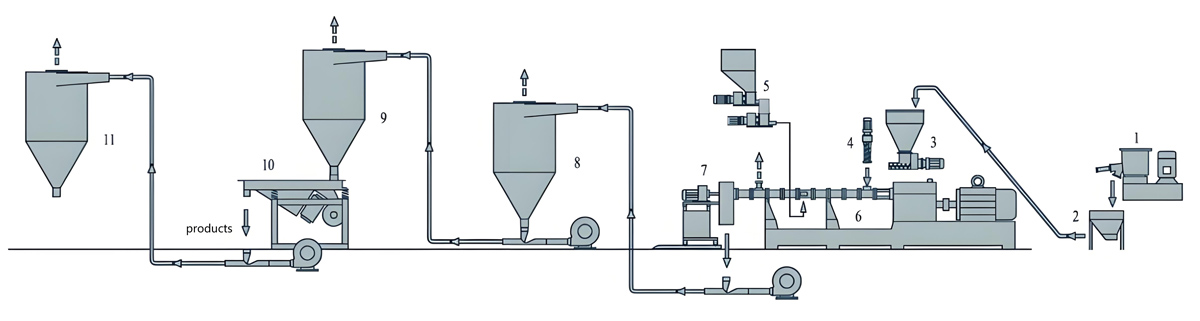
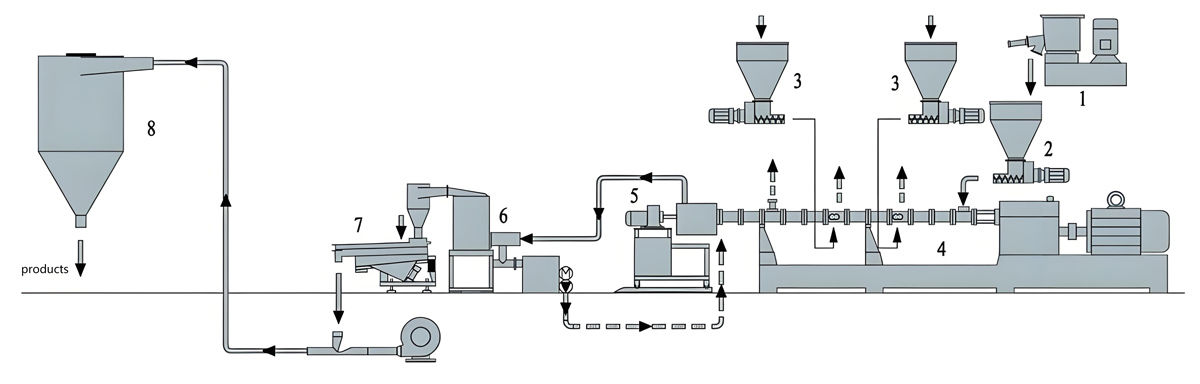
Production Equipment Introduction
Equipment selection for daily chemical packaging masterbatch production addresses quality requirements, regulatory compliance needs, and production efficiency considerations. The Kerke KTE series provides equipment options suitable for various production scale requirements.
Kerke KTE Series for Daily Chemical Packaging
The Kerke KTE series offers equipment options spanning the capacity range required for daily chemical packaging masterbatch production. Equipment selection considers production volume requirements, formulation complexity, and quality verification needs.
The KTE-36B model, with 35.6mm screw diameter and 20 to 100 kg/h throughput, suits pilot production, development activities, and small-scale commercial manufacturing. This equipment enables product development and market testing while maintaining the quality standards daily chemical packaging applications demand.
The KTE-50B model, featuring 50.5mm screw diameter and 80 to 200 kg/h throughput capability, addresses small to medium-scale production requirements. The capacity range supports regional daily chemical packaging masterbatch service with meaningful production volumes.
The KTE-65B, with 62.4mm screw diameter and 200 to 450 kg/h throughput, serves medium-scale production operations with established customer bases. This equipment suits dedicated production facilities serving consistent daily chemical packaging requirements.
The KTE-75B, featuring 71mm screw diameter and 300 to 800 kg/h throughput, provides higher capacity for larger operations. This equipment enables significant production volumes while maintaining the quality control necessary for demanding daily chemical packaging applications.
The KTE-95D, the largest model with 93mm screw diameter and 1000 to 2000 kg/h throughput capability, addresses high-volume production requirements for major daily chemical packaging masterbatch operations. This capacity supports large-scale dedicated production facilities.
Supporting Equipment Considerations
Supporting equipment for daily chemical packaging masterbatch includes material handling systems, quality testing instrumentation, and documentation systems that address the comprehensive requirements of regulated manufacturing.
Material handling equipment emphasizes cleanliness and contamination prevention, including closed systems, clean construction materials, and material segregation capabilities. Gravimetric dosing systems provide precise formulation control essential for consistent quality.
Quality testing instrumentation includes color measurement spectrophotometers, migration testing apparatus, and comprehensive chemical characterization capabilities. These systems enable verification of quality and regulatory compliance throughout production operations.
Parameter Settings for Daily Chemical Packaging Masterbatch
Parameter optimization for daily chemical packaging masterbatch ensures consistent quality while maintaining production efficiency. Careful parameter control addresses specific formulation requirements and quality standards.
Screw Configuration Optimization
Screw configuration optimization for daily chemical packaging masterbatch addresses mixing requirements for high-quality dispersion while maintaining throughput capability. Configuration design considers formulation complexity and quality requirements.
Standard configurations typically employ length-to-diameter ratios of 40:1 to 48:1 with compression ratios of 2.5:1 to 3.5:1. Mixing sections incorporate kneading blocks and distributive mixing elements that achieve uniform component distribution without excessive shear stress.
High-pigment-loading formulations may require modified configurations with enhanced mixing sections or reduced throughput to achieve complete dispersion. The specific configuration depends on formulation characteristics and quality requirements.
Temperature Profile Development
Temperature profile development balances processing efficiency with formulation requirements. Higher temperatures reduce melt viscosity and improve mixing efficiency, but may affect heat-sensitive components.
Typical processing temperatures for polypropylene-based daily chemical packaging masterbatch range from 200 to 260 degrees Celsius across barrel zones. Temperature profiles are adjusted according to specific formulation requirements, with heat-sensitive formulations processed at reduced temperatures.
Temperature gradient design ensures progressive melting and controlled processing throughout the extrusion process. Reduced temperature differentials between zones prevent localized overheating while maintaining processing efficiency.
Throughput and Speed Control
Throughput optimization balances production efficiency with quality requirements. Operating throughputs typically range from 60% to 80% of maximum extruder capacity for most formulations, with reduced throughput for complex or demanding applications.
Screw speed optimization similarly balances mixing intensity against residence time requirements. Operating speeds typically range from 200 to 500 RPM depending on equipment size and formulation characteristics.
Equipment Price Guide
Equipment investment for daily chemical packaging masterbatch production considers quality requirements, capacity needs, and regulatory compliance considerations. The Kerke KTE series provides options across the price spectrum.
The KTE-36B, priced from $25,000 to $35,000, suits development and small-scale production for daily chemical packaging applications. This equipment investment enables quality daily chemical packaging masterbatch production for market entry and specialty applications.
The KTE-50B, priced from $40,000 to $60,000, provides intermediate capacity for established small to medium-scale operations. The throughput capability of 80 to 200 kg/h accommodates regional market service.
The KTE-65B, priced from $50,000 to $80,000, addresses medium-scale production requirements with 200 to 450 kg/h throughput. This investment suits operations with established daily chemical packaging customer bases.
The KTE-75B, priced from $70,000 to $100,000, provides higher capacity for larger operations with substantial production volumes. The 300 to 800 kg/h throughput capability supports significant market presence.
The KTE-95D, priced from $120,000 to $200,000, offers maximum capacity for high-volume production facilities. The 1000 to 2000 kg/h throughput capability enables efficient service for major daily chemical packaging customers.
Problems in Production Process and Solutions
Daily chemical packaging masterbatch production presents specific challenges related to quality consistency, regulatory compliance, and chemical compatibility. Addressing these challenges requires systematic understanding and effective resolution approaches.
Color Consistency Issues
Color consistency problems in daily chemical packaging masterbatch may result from raw material variation, processing inconsistency, or formulation control issues. These problems directly impact brand identity and consumer perception, making color consistency essential for market success.
Root cause investigation examines raw material consistency, process parameter stability, and quality control practices. Color measurement data analysis identifies trends and variation sources that require attention. Equipment calibration verification ensures measurement accuracy.
Resolution approaches include enhanced raw material qualification, process control improvements, and quality system enhancements. Statistical process control implementation provides early detection of variation trends before they cause significant quality problems.
Chemical Compatibility Failures
Chemical compatibility failures occur when masterbatch formulations interact adversely with daily chemical products during storage, resulting in color changes, product contamination, or packaging degradation. These failures may cause product recalls and brand damage.
Diagnostic testing evaluates compatibility under actual or simulated storage conditions. Testing protocols should address relevant exposure conditions, including temperature variations, light exposure, and extended storage periods.
Resolution approaches include reformulation with enhanced chemical resistance, additive adjustments that improve compatibility, and barrier approaches that reduce interaction risks. Collaboration with daily chemical manufacturers enables understanding of specific compatibility requirements.
Migration and Regulatory Compliance Issues
Migration testing failures indicate that masterbatch formulations may release substances exceeding regulatory limits during standardized exposure testing. This compliance failure threatens market access and requires immediate investigation and resolution.
Diagnostic testing characterizes specific migrating substances and their migration rates. This information guides reformulation strategies that address specific migration concerns while maintaining formulation performance and regulatory compliance.
Resolution approaches include reformulation with compliant alternatives, reduced additive loadings, and enhanced barrier approaches. Comprehensive testing during product development establishes formulation compliance margins.
Dispersion Quality Problems
Dispersion quality problems manifest as color streaks, specks, or inconsistent coloration in finished packaging products. These quality issues may result from inadequate mixing, formulation problems, or equipment issues.
Resolution approaches include screw configuration modifications, process parameter adjustments, and formulation optimization. Enhanced pre-mixing procedures may improve preliminary distribution that supports final dispersion quality.
Prevention requires systematic process qualification for each formulation, establishing validated parameters that achieve consistent dispersion quality. Regular equipment maintenance prevents quality degradation from worn components.
Maintenance Requirements
Maintenance practices for daily chemical packaging masterbatch production ensure consistent quality and regulatory compliance. Enhanced maintenance requirements reflect the quality standards necessary for demanding daily chemical packaging applications.
Standard Maintenance Operations
Standard maintenance operations follow established schedules that ensure reliable equipment performance. Daily inspections, weekly maintenance activities, and periodic service address equipment needs while preventing quality issues from equipment condition changes.
Daily maintenance includes equipment inspection, cleaning verification, and parameter monitoring. Any deviations from established operating parameters warrant investigation before they affect product quality.
Periodic maintenance encompasses comprehensive equipment inspection, calibration verification, and component service. Regular maintenance documentation supports quality system requirements and enables maintenance planning.
Regulatory Compliance Maintenance
Maintenance practices for daily chemical packaging masterbatch production include specific attention to regulatory compliance requirements. Documentation maintenance, equipment cleaning verification, and material traceability ensure ongoing compliance capabilities.
Equipment cleaning verification ensures absence of contamination from previous production runs or environmental sources. Documentation maintenance preserves quality records required for regulatory compliance and customer audits.
Frequently Asked Questions
What regulatory requirements affect daily chemical packaging masterbatch?
Daily chemical packaging masterbatch must comply with applicable food contact regulations, cosmetic regulations, and regional requirements. US FDA regulations, EU Regulation 10/2011, and REACH requirements commonly affect formulation compliance strategies.
How do I ensure fragrance compatibility for packaging applications?
Fragrance compatibility requires careful material selection, enhanced chemical resistance testing, and formulation optimization. Specialty high-fastness pigments and barrier additives address specific compatibility concerns for demanding fragrance applications.
What testing is required for food contact compliance?
Food contact compliance testing typically includes overall migration testing, specific migration testing for restricted substances, and sensory testing. Testing protocols depend on specific food contact conditions and target market regulations.
How do I select pigments for daily chemical packaging?
Pigment selection requires verification of regulatory compliance, chemical resistance, and light fastness. Specialty high-purity pigments designated for food contact or cosmetic applications provide compliant options with demonstrated performance characteristics.
What quality systems are necessary for daily chemical packaging masterbatch?
Quality systems include raw material qualification, process controls, final product testing, and compliance documentation management. System requirements may be specified by customers or regulatory requirements for target markets.
How do I address regional regulatory variations?
Addressing regional regulatory variations requires understanding specific market requirements, comprehensive compliance documentation, and formulation strategies that address the most restrictive applicable requirements. Technical expertise and regulatory knowledge support efficient compliance programs.
What documentation supports regulatory compliance?
Compliance documentation includes regulatory compliance certificates, food contact authorizations, REACH compliance statements, safety data sheets, and production batch records. Documentation requirements vary by market but typically require comprehensive material and compliance information.
Conclusion
Daily chemical packaging masterbatch production demands technical expertise across multiple dimensions, including formulation design, regulatory compliance, and quality assurance. The combination of aesthetic requirements for brand differentiation, functional requirements for product protection, and regulatory compliance creates complex challenges that masterbatch producers must address comprehensively. Twin screw extrusion technology, exemplified by the Kerke KTE series equipment, provides the processing capabilities necessary for successful daily chemical packaging masterbatch production when combined with appropriate quality systems and regulatory compliance practices.
Success in the daily chemical packaging market requires understanding of diverse regulatory frameworks, commitment to quality consistency, and comprehensive customer service that supports brand success. The market continues evolving with changing consumer preferences, sustainability requirements, and regulatory developments that influence formulation and manufacturing practices.
Investment in appropriate equipment, quality systems, and technical expertise enables manufacturers to serve the demanding daily chemical packaging market effectively. The combination of capable processing technology, rigorous quality control, and responsive technical support positions producers to address the evolving requirements of daily chemical manufacturers while maintaining the quality and compliance standards that this demanding market segment requires.