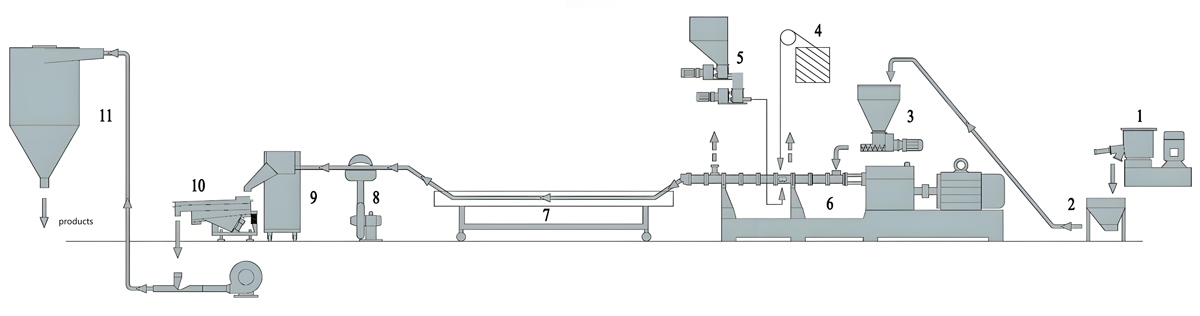
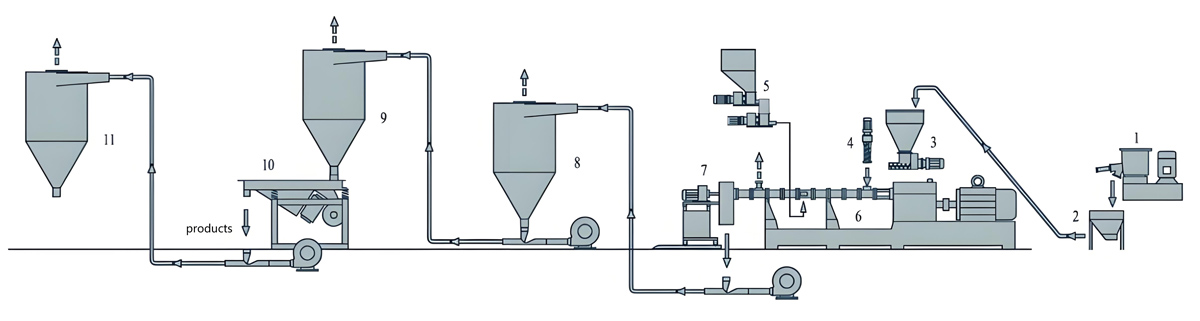
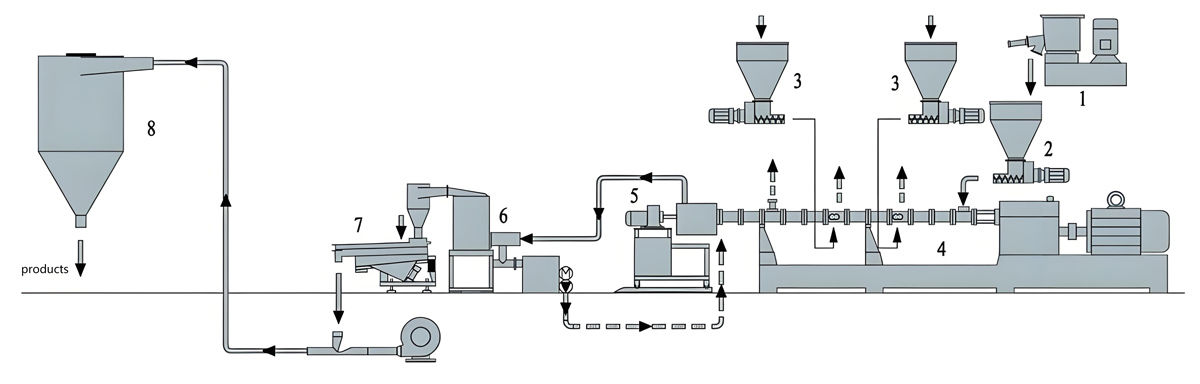
Introduction
Antibacterial masterbatch production represents a rapidly growing segment of the polymer additives industry, driven by increasing demand for hygienic products across medical, food packaging, consumer goods, and public infrastructure applications. The automatic twin screw extruder serves as the essential manufacturing equipment for producing these specialized additive concentrates, offering precise control, automated operation, and consistent quality necessary for delivering effective antibacterial performance while maintaining production efficiency in commercial manufacturing environments.
Antibacterial masterbatches are concentrated formulations containing active antibacterial agents dispersed in carrier resins, designed to provide long-lasting protection against bacterial growth on plastic surfaces. These additives function by incorporating biologically active compounds that inhibit bacterial colonization and reproduction through various mechanisms including membrane disruption, enzyme inhibition, or metabolic interference. Common antibacterial agents include silver-based compounds, zinc pyrithione, quaternary ammonium compounds, and organic antibacterial agents such as triclosan or newer organic alternatives developed for enhanced safety profiles.
The production of antibacterial masterbatch presents unique challenges compared to conventional additive masterbatches, particularly regarding the thermal sensitivity of many antibacterial agents, requirements for precise concentration control given the potency of active ingredients, and the critical importance of achieving uniform dispersion to ensure consistent antibacterial efficacy across all surface areas of final products. Automatic twin screw extruders address these challenges through advanced process control, automated dosing systems, and precise thermal management capabilities that enable consistent production of high-quality antibacterial masterbatches.
This comprehensive guide examines the technical aspects of automatic twin screw extruder applications in antibacterial masterbatch manufacturing, covering formulation considerations, automated production processes, equipment specifications, parameter optimization, quality control requirements, and regulatory considerations essential for successful production in this specialized market segment.
Formulation Ratios for Different Types
Antibacterial masterbatch formulations encompass a diverse range of active agents and concentration levels, each requiring specific formulation approaches to achieve effective antibacterial performance while maintaining processability and compatibility with base polymers. The automatic twin screw extruder must accommodate varied formulation requirements while ensuring precise concentration control and uniform dispersion across different product types.
Silver-based antibacterial masterbatches typically contain 1-5% silver compounds dispersed in appropriate carrier resins. These formulations utilize silver ions or silver nanoparticles as the active antibacterial agent, providing broad-spectrum antibacterial activity with excellent thermal stability and long-term effectiveness. Standard formulations for general-purpose applications might include 2% silver zeolite, 3% silver carrier matrix, and 95% polyethylene carrier resin. The relatively low concentration of active agent demands precise dosing control and exceptional dispersion uniformity to ensure consistent antibacterial coverage in final applications. Silver-based formulations generally offer good thermal stability, allowing processing temperatures up to 260-280°C without significant degradation of antibacterial efficacy.
Zinc-based antibacterial masterbatches incorporate 5-15% zinc compounds such as zinc pyrithione, zinc oxide, or zinc salts dispersed in polymer carriers. These formulations provide effective antibacterial properties with lower material costs compared to silver-based systems. Typical compositions for consumer goods applications might include 10% zinc pyrithione, 5% dispersing agents, and 85% polypropylene carrier resin. The higher active agent concentration requires robust mixing capabilities to achieve uniform distribution, while the thermal stability of zinc compounds allows processing temperatures similar to the carrier resin. Zinc-based formulations often require careful dispersion optimization to prevent aggregation that could reduce antibacterial efficacy or affect product appearance.
Organic antibacterial masterbatches utilize 10-25% organic antibacterial agents such as quaternary ammonium compounds, triclosan derivatives, or newer organic compounds developed for enhanced safety profiles. These formulations are particularly suitable for food contact and medical applications where regulatory approval and safety considerations are critical. A typical formulation might include 15% organic antibacterial agent, 5% compatibility enhancers, and 80% polyethylene or polypropylene carrier resin depending on the intended application. Organic antibacterial agents generally exhibit lower thermal stability compared to inorganic alternatives, requiring careful temperature control during processing to maintain efficacy. Some organic agents may also be sensitive to shear degradation, necessitating optimization of mixing intensity.
Combination antibacterial masterbatches incorporate multiple antibacterial agents to provide synergistic effects and enhanced protection against bacterial strains. These sophisticated formulations typically contain 15-30% total antibacterial content with carefully balanced ratios of different active components. For example, a combination masterbatch for high-performance applications might include 5% silver compound, 10% zinc pyrithione, 5% organic antibacterial agent, and 80% engineering-grade carrier resin. The complexity of these formulations demands advanced screw configurations, precise dosing systems, and sophisticated control algorithms to achieve uniform distribution of all active components while protecting thermally sensitive ingredients.
Specialty antibacterial masterbatches for medical or food contact applications require formulation approaches tailored to meet specific regulatory requirements and performance standards. These formulations often contain 5-20% active agents specifically selected for regulatory approval in sensitive applications. Carrier resins must be carefully selected for compatibility with both base polymers and regulatory requirements, often requiring medical-grade or food-contact-approved materials. Twin screw extruder configurations for these applications typically include enhanced temperature control capabilities, specialized screw designs for gentle processing of sensitive active agents, and advanced cleaning protocols to prevent cross-contamination between different product types.
Production Process
The automated production process for antibacterial masterbatch using twin screw extruders integrates multiple advanced systems to ensure precise concentration control, uniform dispersion, and consistent product quality. The automation capabilities of modern twin screw extruders enable continuous monitoring and adjustment of critical parameters, reducing operator intervention and improving production consistency compared to manual or semi-automated systems.
Automated raw material handling and dosing systems establish the foundation for precise concentration control in antibacterial masterbatch production. Gravimetric dosing systems with loss-in-weight technology provide continuous monitoring and adjustment of feed rates for all formulation components, ensuring that active antibacterial agents are maintained at precise concentrations throughout the production run. These systems typically include separate hoppers for carrier resin and active ingredients, each equipped with load cells for continuous weight monitoring. The control system automatically adjusts feeder screw speeds based on real-time weight loss measurements, compensating for variations in material density or flow characteristics that could otherwise cause concentration drift. For multi-component formulations, multiple dosing systems operate in coordinated control to maintain proper ratios between all components.
Material feeding and introduction to the extruder barrel is automated through precise control of feeders and timing systems. Main feeders introduce bulk carrier resin and high-concentration premixes through the primary feed port, while side feeders may be employed for temperature-sensitive antibacterial agents that require introduction downstream of initial heating zones. Liquid dosing systems with heated lines and metering pumps precisely deliver liquid antibacterial agents at specified injection points. The automation system synchronizes all feeding operations with extruder operating conditions, adjusting feed rates based on screw speed, barrel fill level, and process conditions to maintain optimal processing conditions and consistent product quality.
Plasticization and melting operations in automatic twin screw extruders benefit from precise temperature control and automated adjustment capabilities. Temperature profiles are established based on formulation thermal characteristics and antibacterial agent stability requirements. The control system continuously monitors multiple barrel zones, adjusting heating and cooling output to maintain setpoints despite variations in ambient conditions or process disturbances. For formulations containing thermally sensitive organic antibacterial agents, the system can implement specialized temperature profiles with gradual heating through transition zones and careful temperature control in mixing zones to prevent degradation. Automated temperature ramping during startup and controlled cooling during shutdown protect sensitive components from thermal shock that could affect antibacterial efficacy.
Automated mixing and dispersion operations utilize advanced screw designs and process control algorithms to achieve uniform distribution of antibacterial agents. The automation system monitors key mixing parameters including torque, pressure, and temperature profiles, automatically adjusting screw speed and throughput to maintain optimal mixing conditions. For formulations requiring specific shear profiles, the system can implement variable screw speed programs that adjust mixing intensity based on the specific requirements of different formulation components. Real-time monitoring of energy consumption and mixing efficiency enables the system to detect deviations from optimal mixing conditions and implement corrective adjustments before product quality is affected.
In-process quality monitoring and control systems represent a significant advantage of automatic twin screw extruders for antibacterial masterbatch production. Near-infrared spectroscopy systems can monitor active agent concentration in real-time, providing immediate feedback for dosing system adjustments. Rheological monitoring systems track melt viscosity, detecting variations that could indicate concentration changes or dispersion issues. Inline particle size analysis systems can verify dispersion quality, detecting agglomerates before they affect final product quality. The automation system integrates all monitoring data, implementing automatic adjustments or alerting operators to intervene when parameters deviate from acceptable ranges.
Automated pelletizing and packaging operations complete the production process with minimal operator intervention. Strand pelletizers with automatic knife adjustment systems maintain consistent pellet size despite variations in strand characteristics. Underwater pelletizing systems with automated cutter gap adjustment produce uniform pellets with minimal fines. Automated packaging systems weigh and package finished masterbatch into bags or containers with precise weight control and batch tracking capabilities. The entire production line operates under integrated control, enabling continuous operation with consistent quality output and minimal manual intervention.
Production Equipment Introduction
Modern automatic twin screw extruders for antibacterial masterbatch production incorporate advanced automation features specifically designed to address the unique requirements of producing these high-value, performance-critical additives. The integration of automated dosing, process control, and quality monitoring systems enables consistent production of masterbatches with precise concentration control and uniform dispersion, essential for reliable antibacterial performance in final applications.
Nanjing Kerke Extrusion Equipment Company KTE Series automatic twin screw extruders provide comprehensive solutions for antibacterial masterbatch production, combining proven twin screw extruder technology with advanced automation capabilities. These systems feature co-rotating screw designs with L/D ratios ranging from 40:1 to 48:1, providing the extended residence time necessary for achieving uniform dispersion while maintaining production throughput that meets commercial requirements. The integrated control architecture enables seamless coordination between extruder operation, dosing systems, and quality monitoring equipment, creating a fully automated production cell requiring minimal operator intervention for routine operation.
Automated dosing and feeding systems represent critical components for antibacterial masterbatch production, where precise concentration control is essential given the potency and cost of active antibacterial agents. The KTE Series extruders can be equipped with multiple gravimetric dosing systems featuring loss-in-weight technology for continuous weight monitoring and automatic feed rate adjustment. These systems typically include separate feeders for carrier resin and active antibacterial agents, each with independent control while maintaining coordinated operation. For liquid antibacterial agents, precision metering pumps with heated lines and flow monitoring ensure accurate delivery at specified injection points. Side feeding capabilities allow temperature-sensitive agents to be introduced downstream of initial heating zones, minimizing thermal exposure and preserving antibacterial efficacy.
Advanced temperature control systems provide the precise thermal management required for thermally sensitive antibacterial agents. The KTE Series extruders feature electrically heated barrels with 8-12 independent temperature control zones, each equipped with PID controllers and rapid-response cooling systems for precise temperature regulation. The control system can implement complex temperature profiles with different setpoints for each zone, enabling gradual heating through transition zones and reduced temperatures in mixing zones for formulations containing thermally sensitive components. Automated temperature ramping during startup and controlled cooling during shutdown protect sensitive components from thermal stress that could affect antibacterial performance.
Process monitoring and control systems provide comprehensive oversight of all production parameters and enable automatic adjustment to maintain optimal conditions. The KTE Series extruders feature touchscreen HMI interfaces with real-time display of critical parameters including temperatures, screw speed, feed rates, torque, pressure, and motor load. Advanced control algorithms implement automatic regulation of temperature, pressure, and throughput, maintaining stable operating conditions despite external disturbances. Data logging capabilities record all process parameters with timestamps, providing complete traceability for quality assurance and regulatory compliance. Remote monitoring capabilities enable supervisory oversight and quick response to process deviations.
In-line quality monitoring systems can be integrated with the extruder control system to provide real-time assessment of product quality and enable immediate corrective actions. Near-infrared spectroscopy systems mounted on the extruder die can measure active agent concentration in the melt stream, providing continuous concentration verification. Rheological monitoring systems track melt viscosity, detecting variations that could indicate formulation or processing issues. Inline microscopy or particle size analysis systems can evaluate dispersion quality, detecting agglomerates or inhomogeneities. The control system integrates this monitoring data, implementing automatic adjustments to dosing systems or process parameters to maintain product quality specifications.
Drive systems and power management for automatic twin screw extruders provide consistent performance while optimizing energy consumption. The KTE Series extruders are equipped with high-efficiency AC drive motors with power ratings from 55-250 kW depending on extruder size and production requirements. Torque capabilities typically range from 500-1500 Nm, providing sufficient power for intensive mixing operations required for uniform dispersion. Variable frequency drives enable precise speed control and optimize energy consumption by matching motor output to actual load requirements. Power monitoring systems track energy consumption and can implement energy-saving strategies such as reduced speed during low-demand periods or automatic shutdown during idle periods.
Downstream equipment for automatic antibacterial masterbatch production includes automated pelletizing systems, quality inspection equipment, and packaging systems that complete the automated production line. Strand pelletizers with automatic knife adjustment and wear compensation maintain consistent pellet size without manual intervention. Underwater pelletizing systems with automated process control produce uniform pellets with excellent surface finish. Quality inspection systems including automated size classification, color monitoring, and metal detection ensure that finished product meets specifications before packaging. Automated packaging systems weigh, fill, and seal bags or containers with precise weight control and complete batch tracking for traceability requirements.
Parameter Settings
Optimization of process parameters for automatic twin screw extruder production of antibacterial masterbatch requires systematic evaluation of multiple variables while leveraging automation capabilities to maintain precise control. The integrated control system enables precise management of temperature profiles, screw speed, feed rates, and mixing intensity to achieve optimal balance between antibacterial agent dispersion, thermal stability, and production efficiency.
Temperature profile optimization represents one of the most critical aspects of antibacterial masterbatch production, particularly for formulations containing thermally sensitive organic antibacterial agents. For polyethylene-based formulations containing silver compounds, a typical temperature profile might include feed zone at 150-160°C, first transition zone at 160-170°C, second transition zone at 170-180°C, mixing zone 1 at 180-190°C, mixing zone 2 at 190-200°C, metering zone at 200-210°C, and die zone at 210-220°C. Formulations containing organic antibacterial agents may require reduced temperatures, particularly in mixing zones, with typical settings 20-30°C lower to protect thermally sensitive components. The automation system can implement precise temperature control across all zones and can adjust temperatures based on real-time monitoring of antibacterial agent stability indicators.
Screw speed selection impacts mixing intensity, residence time, and shear exposure for antibacterial agents. Higher screw speeds generate increased shear forces that improve dispersion but may also increase thermal load on sensitive components. Typical operating speeds for antibacterial masterbatch range from 100-300 RPM, with lower speeds (100-150 RPM) appropriate for formulations containing highly thermally sensitive organic agents, and higher speeds (200-300 RPM) suitable for formulations based on more thermally stable inorganic compounds like silver or zinc derivatives. The automation system can implement variable screw speed programs that adjust speed based on the specific requirements of different formulation zones, using lower speeds during initial melting and higher speeds during mixing phases to optimize dispersion while protecting sensitive components.
Feed rate and throughput optimization directly impacts production efficiency and product quality consistency. The gravimetric dosing systems automatically adjust feed rates to maintain precise concentration control, but the overall throughput must be optimized based on formulation characteristics and equipment capacity. For antibacterial masterbatch production, throughput rates typically range from 80-400 kg/hour depending on extruder size and formulation complexity. The automation system maintains optimal fill level in the extruder barrel, typically 70-85% of maximum volumetric capacity, by coordinating feed rates with screw speed and processing conditions. This coordinated control ensures consistent residence time and mixing efficiency while maximizing production efficiency.
Mixing element configuration significantly affects dispersion quality and antibacterial agent distribution uniformity. For antibacterial masterbatch production, screw configurations typically include multiple mixing zones with different mixing element types. Initial mixing zones may include mild distributive mixing elements to begin dispersing active agents without excessive shear that could degrade sensitive components. Subsequent mixing zones may include more intensive dispersive elements such as kneading blocks to break down any remaining agglomerates and achieve uniform distribution. The automation system can monitor mixing efficiency through torque and energy consumption measurements, automatically adjusting screw speed or throughput to maintain optimal mixing conditions.
Concentration control parameters are critical for antibacterial masterbatch given the potency and cost of active agents. The gravimetric dosing systems provide continuous concentration monitoring and automatic adjustment to maintain target concentrations within tight tolerances, typically ±1-2% of target concentration. The control system coordinates multiple dosing systems for multi-component formulations, maintaining proper ratios between all active agents and carrier components. Real-time concentration monitoring through inline spectroscopy provides immediate feedback for dosing adjustments, ensuring that concentration deviations are detected and corrected before they affect product quality.
Die temperature and pressure control parameters directly impact strand formation and pellet quality. Die temperature should be maintained 5-10°C above the melt temperature to ensure smooth flow, with the automation system automatically adjusting die heating based on melt temperature measurements. Die pressure typically ranges from 2-5 MPa for antibacterial masterbatch production, with the control system monitoring pressure and implementing automatic adjustments to feed rate or screw speed to maintain stable pressure. Pressure fluctuations can indicate concentration changes or processing issues, with the system triggering alarms or automatic adjustments when pressure exceeds specified limits.
Cooling and solidification parameters significantly affect pellet quality and dimensional stability. Water bath temperature should be maintained between 30-50°C for most antibacterial masterbatch formulations, with the automation system implementing precise temperature control to maintain consistent cooling rates. Bath immersion length must be optimized to ensure complete solidification, typically 3-5 meters depending on line speed and formulation characteristics. The automation system can adjust water bath circulation rate and temperature based on line speed and ambient conditions to maintain consistent cooling. For formulations containing components with specific crystallinity requirements, the system can implement controlled cooling profiles to optimize solidification and pellet properties.
Equipment Price
Investment in automatic twin screw extruder equipment for antibacterial masterbatch production encompasses multiple cost categories including the base extruder system, automation components, quality monitoring equipment, and integration services. Understanding the cost structure and value propositions of different automation levels enables informed investment decisions aligned with production requirements, quality standards, and business objectives.
Basic automated twin screw extruder systems for antibacterial masterbatch production typically range from 120,000-200,000 USD. These systems include the base extruder with standard automation features including PLC-based control, touchscreen HMI, basic temperature control, and single gravimetric dosing system for carrier resin. While these systems provide some level of automation, they may require operator intervention for parameter adjustments, quality monitoring, and troubleshooting. These systems are suitable for producers with moderate production volumes who require automation benefits but have limited capital budgets or less stringent quality requirements.
Mid-range automatic systems such as the Nanjing Kerke KTE Series with advanced automation features typically represent investments of 200,000-350,000 USD. These systems include comprehensive automation capabilities including multiple gravimetric dosing systems for precise concentration control, advanced multi-zone temperature control with precise regulation, integrated process monitoring with real-time parameter display and data logging, and automated pelletizing systems. These systems provide the automation level necessary for commercial-scale production of antibacterial masterbatches with consistent quality and minimal operator intervention. The enhanced automation reduces labor requirements, improves quality consistency, and provides traceability documentation valuable for regulatory compliance.
Advanced fully automated production systems for large-scale antibacterial masterbatch manufacturing typically range from 400,000-700,000 USD or more depending on specifications and integration level. These systems feature the highest level of automation including in-line quality monitoring systems such as NIR spectroscopy and rheological monitoring, advanced process control with automatic parameter adjustment, comprehensive data acquisition and storage for quality assurance, and full integration of dosing, extrusion, pelletizing, and packaging operations. These systems provide maximum production efficiency with minimal operator intervention, exceptional quality consistency, and complete batch traceability essential for medical or food-contact applications with stringent regulatory requirements.
Automation components and quality monitoring equipment significantly impact total investment. Additional gravimetric dosing systems for multi-component formulations typically cost 25,000-50,000 USD each depending on capacity and precision requirements. In-line NIR spectroscopy systems for concentration monitoring cost approximately 30,000-60,000 USD including installation and calibration. Rheological monitoring systems for melt viscosity tracking cost 20,000-40,000 USD. Automated pelletizing systems with advanced features add 30,000-60,000 USD. Integration services including programming, commissioning, and operator training typically cost 15-25% of equipment value depending on system complexity and integration requirements.
Operational cost considerations for automatic systems include energy consumption, maintenance requirements, labor costs, and quality-related expenses. Automated systems typically have higher initial costs but lower operational costs due to reduced labor requirements and improved consistency reducing scrap and rework. Energy consumption varies based on system size and operating parameters, with automatic systems often optimizing energy consumption through smart control algorithms. Maintenance costs include regular servicing of automation components, calibration of sensors and instruments, and software updates. The improved consistency and reduced operator error in automatic systems typically result in lower quality-related costs including reduced scrap rates, fewer customer complaints, and reduced need for retesting.
Total cost of ownership analysis should consider factors beyond initial investment including production capacity, quality requirements, regulatory compliance needs, and expected equipment service life. Higher levels of automation may justify their increased cost through improved product quality, reduced labor costs, enhanced regulatory compliance capabilities, and increased production efficiency. Financing options including equipment leasing, vendor financing programs, and government incentives for automation can help manage capital requirements. Used equipment options may provide cost savings but typically offer limited automation capabilities compared to new equipment and may require significant upgrades to meet current automation standards.
Production Problems and Solutions
Problem 1: Inconsistent Antibacterial Agent Concentration
Problem Analysis: Inconsistent antibacterial agent concentration results in variable product performance and potential failure to meet customer specifications or regulatory requirements. This issue manifests as concentration variations between batches, concentration drift during production runs, or non-uniform distribution within pellets. Given the potency of antibacterial agents, even small concentration variations can significantly affect product efficacy. Root causes include feeding system inaccuracies, material density variations, formulation component segregation, or process parameter variations that affect distribution uniformity.
Causes: Volumetric feeding systems lack sufficient accuracy; gravimetric feeders not properly calibrated; material density variations affect feed rate; improper pre-mixing leads to component segregation; screw speed or throughput variations change residence time; temperature variations affect material viscosity and flow; worn feeding components reduce accuracy; lack of in-line concentration monitoring allows deviations to persist.
Solutions: Implement multiple gravimetric dosing systems with loss-in-weight technology for all active components; ensure proper pre-mixing of formulation components before feeding; calibrate feeding systems regularly; implement automatic feeding coordination with process parameters; install in-line concentration monitoring using NIR spectroscopy; use statistical process control to detect concentration trends; replace worn feeding components promptly; implement automated feedback control between concentration monitoring and dosing systems.
Prevention Methods: Establish strict material handling and storage procedures; implement comprehensive preventive maintenance for feeding systems; develop and implement analytical testing protocols for concentration verification; use in-line monitoring with automatic feedback control; train operators on proper feeding system operation and maintenance; maintain detailed production records for traceability; implement automated recipe management systems to ensure correct parameter settings for each formulation.
Problem 2: Thermal Degradation of Antibacterial Agents
Problem Analysis: Thermal degradation of antibacterial agents reduces or eliminates antibacterial efficacy, potentially resulting in products that fail to provide required protection against bacterial growth. This issue is particularly critical for organic antibacterial agents with limited thermal stability. Degradation typically occurs when local temperatures exceed thermal stability limits due to excessive heating, inadequate temperature control, extended residence times at elevated temperatures, or hot spots in processing equipment. Even minor degradation can significantly reduce efficacy given the low concentrations typically used.
Causes: Barrel temperatures set too high for thermal stability limits; screw speed generates excessive shear heating; inadequate barrel cooling; residence time too long; worn screw elements increase friction and heat generation; temperature sensors inaccurate; die temperature too high; lack of thermal stability data for formulation components.
Solutions: Reduce barrel temperatures in critical zones; lower screw speed while maintaining adequate dispersion; verify proper operation of barrel cooling systems; increase throughput rate to reduce residence time; replace worn screw elements; calibrate temperature sensors; optimize die temperature; implement side feeding for thermally sensitive components; conduct thermal analysis to establish safe processing windows.
Prevention Methods: Establish strict temperature limits based on thermal analysis data; install additional temperature sensors for hot spot monitoring; implement temperature interlocks preventing operation above safe limits; conduct regular thermal analysis on product samples; train operators on recognizing degradation symptoms; develop formulations with enhanced thermal stability; use automated temperature control systems with safety limits.
Problem 3: Incomplete Dispersion of Antibacterial Agents
Problem Analysis: Incomplete dispersion of antibacterial agents results in uneven distribution throughout the carrier resin, causing inconsistent antibacterial performance across product surfaces and potential failure to provide uniform protection. This issue manifests as visible particles or specks, variable efficacy in testing, or customer complaints about inconsistent performance. Root causes include insufficient mixing intensity, inadequate residence time, inappropriate screw configuration, or formulation characteristics that resist dispersion. Given the low concentrations used, uniform dispersion is critical for consistent performance.
Causes: Screw speed too low for adequate dispersion; mixing zones insufficient for formulation requirements; residence time too short; screw configuration not optimized for dispersion; feed rate too high for mixing capacity; particle size of antibacterial agents too large; worn mixing elements reduce shear efficiency; formulation components incompatible causing aggregation.
Solutions: Increase screw speed within thermal stability limits; modify screw configuration to include additional mixing elements; reduce throughput to increase residence time; optimize mixing element types and placement; pre-grind antibacterial agents to appropriate particle size; replace worn mixing elements; evaluate formulation compatibility; implement inline dispersion monitoring.
Prevention Methods: Develop formulation-specific screw configurations; establish minimum mixing criteria for each formulation; implement regular dispersion testing; monitor mixing efficiency through torque and energy consumption; maintain screw elements in optimal condition; use pre-dispersed antibacterial agent concentrates when available; implement statistical process control on dispersion quality parameters.
Problem 4: Cross-Contamination Between Product Types
Problem Analysis: Cross-contamination between different antibacterial masterbatch types can cause performance issues, regulatory non-compliance, and customer complaints. This issue occurs when residue from previous production runs contaminates subsequent batches, particularly problematic when switching between products containing different active agents or between antibacterial and non-antibacterial products. Given the potency and potential regulatory implications of cross-contamination, thorough cleaning between product changes is essential for quality assurance.
Causes: Inadequate cleaning procedures between product changes; equipment design features that trap material; similar product properties preventing visual detection of contamination; lack of cleaning verification protocols; operator error in cleaning procedures; insufficient purging material volume; complex equipment geometry that resists cleaning.
Solutions: Develop detailed cleaning procedures for each product change sequence; use dedicated equipment for product families when possible; implement cleaning verification testing; use appropriate purging materials and volumes; train operators on cleaning procedures; redesign equipment to eliminate material traps; implement automated cleaning systems when feasible.
Prevention Methods: Establish cleaning validation protocols for product changeovers; implement batch tracking and traceability systems; schedule production runs to minimize changeover frequency; use color or visual indicators to detect cross-contamination; maintain dedicated tooling for different product types; implement regular equipment inspection to detect material buildup; train operators on cross-contamination risks and prevention.
Problem 5: Automation System Failures
Problem Analysis: Automation system failures can cause production interruptions, quality variations, and safety issues if not properly managed. These failures may occur in control systems, sensors, dosing systems, or communication networks. The impact ranges from minor parameter deviations requiring manual intervention to complete system shutdowns halting production. Given the critical role of automation in maintaining precise concentration control and consistent quality, system reliability is essential for commercial production.
Causes: Software bugs or corruption; hardware component failures; electrical power disturbances; communication network failures; sensor calibration drift; improper maintenance; environmental factors affecting electronics; inadequate backup systems.
Solutions: Implement redundant control systems for critical functions; maintain backup power supplies; perform regular software updates and maintenance; implement predictive maintenance on critical components; establish emergency operating procedures for manual operation; train operators on manual override procedures; implement regular system diagnostics and testing.
Prevention Methods: Establish comprehensive preventive maintenance program for automation systems; implement regular calibration schedules for sensors and instruments; maintain updated software and firmware; train maintenance personnel on automation system troubleshooting; implement system redundancy for critical functions; establish regular backup procedures for programs and data; document all system configurations and changes.
Maintenance and Care
Comprehensive maintenance programs for automatic twin screw extruders used in antibacterial masterbatch production are essential for ensuring consistent product quality, maximizing production uptime, and protecting the substantial investment represented by automation systems. The maintenance program must address not only the mechanical extruder components but also the automation systems, sensors, and control equipment that enable automated operation.
Daily maintenance procedures should be performed at the start of each production shift to identify potential issues before they cause production interruptions. These procedures include visual inspection of all mechanical components for damage or abnormalities; verification of all safety interlocks and emergency stop functionality; checking automation system status including PLC operation, HMI displays, and communication networks; verifying proper operation of all temperature control zones; inspecting dosing systems for proper operation and consistent material flow; monitoring drive system operation for unusual sounds or vibrations; and verification of proper operation of all sensors and monitoring instruments. Documenting these daily checks creates a maintenance history that helps identify developing issues before they cause equipment failure.
Weekly maintenance tasks address automation components and precision equipment that require regular attention. These tasks include cleaning and inspecting all sensor elements including temperature probes, pressure transmitters, and load cells; verifying calibration accuracy of critical sensors; checking and tightening all electrical connections and terminal boards; inspecting and cleaning dosing system components including feeder screws, hoppers, and transfer lines; verifying proper operation of all pneumatic and hydraulic systems; cleaning HMI screens and checking for proper operation; inspecting and cleaning pelletizer components; and verifying proper operation of all communication networks and data logging systems. Weekly maintenance ensures that automation components maintain their accuracy and reliability.
Monthly maintenance procedures include more detailed inspection and testing of automation and mechanical systems. These tasks include comprehensive testing of all safety systems and interlocks; calibration of all sensors and instruments against reference standards; detailed inspection of dosing system accuracy using verification procedures; testing backup systems and redundancy features; reviewing and analyzing system logs for error patterns or trends; inspecting electrical cabinets for proper cleanliness and ventilation; testing all manual override functions to ensure proper operation when needed; and reviewing and updating software backups and documentation. Monthly maintenance provides early detection of performance degradation and ensures that backup systems are functional when needed.
Quarterly maintenance encompasses comprehensive calibration and preventive replacement activities. These tasks include complete calibration of all temperature sensors and control loops; calibration of all dosing systems and flow meters; detailed inspection and testing of PLC and control system hardware; testing of all communication networks and data logging systems; inspection and replacement of wearing components in dosing systems including feeder screws and bearings; comprehensive electrical system inspection including testing of motor drives, power distribution, and grounding systems; detailed review of system performance data to identify trends or developing issues; and software updates and system optimization based on manufacturer recommendations. Quarterly maintenance ensures that systems maintain peak performance and reliability.
Annual maintenance represents the most comprehensive maintenance activities and should include complete system audits and overhauls as needed. These tasks include complete system backup and reinstallation of control software; detailed inspection and testing of all electrical and electronic components; comprehensive calibration of all instruments and sensors; complete overhaul of dosing systems including replacement of worn components; detailed inspection of mechanical components including screws, barrels, and drives; review and update of all documentation including maintenance procedures and system manuals; comprehensive testing of all system functions and backup procedures; and detailed review of maintenance records to identify trends or recurring issues. Annual maintenance provides the opportunity for major system updates and optimizations.
Automation system maintenance represents a critical component given the role of automation in maintaining product quality and production efficiency. This includes regular software maintenance including updates, patches, and virus protection; database maintenance including data archiving and cleanup; network maintenance including bandwidth optimization and security updates; and cybersecurity maintenance including vulnerability assessments and security updates. The increasing connectivity of automation systems requires attention to cybersecurity to protect against unauthorized access or malicious attacks that could compromise production or quality.
Maintenance record-keeping and analysis provides valuable information for optimizing maintenance intervals, predicting failures before they occur, and identifying opportunities for system improvements. Comprehensive records should include dates and details of all maintenance activities, calibration results with before/after values, system error logs and resolution details, software changes and version numbers, spare parts usage, and any production issues related to equipment or automation problems. Analysis of these records enables predictive maintenance strategies that replace components based on condition and performance trends rather than fixed intervals, optimizing maintenance costs and minimizing unplanned downtime.
FAQ
What level of automation is appropriate for different production volumes in antibacterial masterbatch manufacturing?
The appropriate level of automation depends on production volume, quality requirements, and regulatory compliance needs. For pilot-scale or low-volume production below 100 kg/day, basic automation with PLC control and single gravimetric dosing may be sufficient. For medium-scale production up to 500 kg/day, advanced automation including multiple dosing systems and process monitoring provides significant benefits in quality consistency and labor efficiency. For large-scale production above 500 kg/day or for medical and food-contact applications with stringent regulatory requirements, fully automated systems with in-line quality monitoring, advanced process control, and comprehensive data acquisition are typically justified by the improved quality, reduced risk of regulatory non-compliance, and lower labor costs.
How do in-line monitoring systems improve quality control in antibacterial masterbatch production?
In-line monitoring systems provide real-time assessment of critical quality attributes, enabling immediate detection and correction of issues before they affect finished product. NIR spectroscopy systems monitor antibacterial agent concentration continuously, catching concentration drifts that would otherwise go undetected until offline testing revealed problems. Rheological monitoring tracks melt viscosity, detecting variations that could indicate formulation changes or processing issues. Inline dispersion monitoring can detect agglomerates or inhomogeneities before they reach the finished product. This real-time monitoring significantly reduces the risk of producing non-conforming material, reduces scrap rates, and provides documentation for regulatory compliance and customer quality assurance requirements.
What cleaning procedures are necessary when switching between different antibacterial masterbatch products?
Cleaning procedures for product changeovers depend on the similarity between products and regulatory requirements. For products with similar carrier resins and compatible active agents, purging with 2-3 times the extruder volume of carrier resin may be sufficient. For products with different carrier resins or incompatible active agents, more extensive cleaning including mechanical cleaning of hoppers, feed lines, and die areas may be required. For changes between antibacterial and non-antibacterial products, or between products for different regulatory applications, complete equipment disassembly and cleaning may be necessary to prevent cross-contamination. Cleaning verification should include visual inspection, analytical testing for active agents, and production of test batches to confirm absence of cross-contamination.
How can I validate the effectiveness of antibacterial masterbatch produced with twin screw extruders?
Validation of antibacterial effectiveness requires comprehensive testing following established protocols such as ISO 22196 for antibacterial activity on plastic surfaces, JIS Z 2801, or ASTM E2149 for antibacterial activity under dynamic contact conditions. Testing should include verification of antibacterial efficacy against relevant bacterial strains, measurement of minimum inhibitory concentration, evaluation of longevity of antibacterial effect over time, and assessment of performance after simulated aging or use conditions. Production validation should include testing of multiple batches to confirm consistency, testing of samples from different production runs, and verification that the masterbatch provides the specified antibacterial performance when incorporated into final products at recommended dosage levels.
What regulatory considerations are important when producing antibacterial masterbatch?
Regulatory considerations for antibacterial masterbatch production depend on the intended end-use applications. For medical device applications, compliance with FDA regulations, EU Medical Device Regulation, and relevant ISO standards is required. For food contact applications, compliance with FDA food contact regulations, EU Regulation 10/2011, and other regional requirements is essential. For general consumer applications, compliance with EPA regulations for antimicrobial products and relevant local regulations may be required. Producers must maintain comprehensive documentation including raw material specifications, production records, test data, and traceability information. Quality systems meeting ISO 9001 or other relevant standards provide frameworks for ensuring compliance with regulatory requirements.
How do I optimize the balance between antibacterial efficacy and cost in masterbatch formulations?
Optimizing the balance between efficacy and cost requires systematic evaluation of antibacterial agents at different concentration levels and correlation with performance testing. Testing should identify minimum effective concentrations for required performance against target bacterial strains. Cost-benefit analysis should consider not only raw material costs but also processing requirements, dispersion challenges, and performance longevity. Selection of antibacterial agents should consider factors beyond initial cost including thermal stability during processing, compatibility with carrier resins, and regulatory approval status. The optimization process typically involves iterative testing of formulations with different agents and concentrations, measuring performance against both technical requirements and cost targets to identify the optimal balance point.
What are the most common causes of antibacterial agent degradation during processing?
The most common causes of antibacterial agent degradation include thermal exposure above the thermal stability limit, shear degradation from excessive mixing intensity, chemical reactions with other formulation components, and oxidation during high-temperature processing. Thermal degradation is particularly problematic for organic antibacterial agents with limited thermal stability. Shear degradation can occur in high-shear mixing zones, especially for compounds with large molecular structures. Chemical reactions can occur when incompatible formulation components are processed together, potentially neutralizing antibacterial activity. Oxidation can affect certain organic compounds, especially when processed at high temperatures for extended periods. Prevention requires careful temperature control, optimization of mixing intensity, formulation compatibility testing, and use of stabilizers when appropriate.
How can I ensure traceability of antibacterial masterbatch production for regulatory compliance?
Ensuring traceability requires comprehensive documentation and tracking systems that follow each batch from raw materials through production to final delivery. Key elements include unique batch identification for each production run, complete records of all raw materials used with lot numbers and certificates of analysis, detailed production records including all process parameters and timestamps, quality testing results with sample identification, and shipping records linking final product to production batches. Automated systems with integrated data logging and barcode or RFID tracking significantly enhance traceability capabilities. Regular audits of traceability systems ensure that records are complete, accurate, and readily available for regulatory inspections or customer inquiries.
Conclusion
The production of antibacterial masterbatch using automatic twin screw extruder technology represents a sophisticated manufacturing process that demands integration of advanced automation capabilities with specialized knowledge of antibacterial agent chemistry and processing requirements. The increasing market demand for hygienic products across diverse applications creates significant opportunities for producers who can deliver consistent quality while managing the technical challenges associated with these specialized additives.
Automatic twin screw extruders provide the technological foundation for successful antibacterial masterbatch production, offering the precise control, automated operation, and quality monitoring capabilities necessary to meet the exacting requirements of this market segment. Equipment such as the Nanjing Kerke KTE Series with integrated dosing systems, advanced process control, and in-line quality monitoring enables consistent production of masterbatches with precise concentration control and uniform dispersion, essential for reliable antibacterial performance in final applications.
The complexity of antibacterial masterbatch production extends beyond simple concentration accuracy to include dispersion uniformity, thermal stability of active agents, prevention of cross-contamination, and compliance with regulatory requirements for different end-use applications. Achieving these quality standards demands comprehensive automation systems that minimize human error, provide real-time quality monitoring, and maintain detailed traceability documentation. The most successful producers invest in advanced automation not just for labor reduction but for quality assurance and regulatory compliance capabilities.
Looking forward, the antibacterial masterbatch market will continue evolving with new antibacterial agent technologies, enhanced performance requirements, increasingly stringent regulatory standards, and growing demand for sustainable and environmentally responsible formulations. Successful producers will continue investing in advanced extrusion technology, automation capabilities, and quality control systems to maintain competitive advantage. The fundamental principles of precise dosing, uniform dispersion, and thermal management will remain essential, but their application will evolve with new technologies and market requirements.
By implementing the technical principles and best practices outlined in this comprehensive guide, producers can optimize their automatic twin screw extruder operations for antibacterial masterbatch manufacturing, achieve superior product quality and consistency, meet regulatory compliance requirements, and establish strong positions in this growing and technically demanding market segment. The integration of appropriate automation technology, formulation expertise, and quality assurance creates the foundation for sustainable success in antibacterial masterbatch production.