Introduction
Food grade masterbatch production represents a specialized segment of the compounding industry, serving critical applications including food packaging, beverage containers, food processing equipment, and consumer products that come into contact with food. The production of food grade masterbatches requires equipment capable of meeting stringent regulatory requirements for food contact materials while maintaining consistent product quality and performance. Co-rotating twin screw extruders have become essential equipment for this application due to their superior mixing capabilities, self-cleaning characteristics, and ability to meet food contact regulations. The integration of sanitary design features in co-rotating twin screw extruders addresses the demanding requirements of food contact material production while maintaining the mixing performance necessary for high-quality masterbatch production.
The significance of food grade masterbatches extends beyond regulatory compliance to include considerations of color stability, migration limits, and organoleptic properties. These masterbatches typically contain colorants, functional additives, and carrier polymers that must meet food contact regulations including FDA, EU, and other international standards. The formulations often use colorants approved for food contact and additives with low migration characteristics. The production process must prevent contamination from previous production runs and ensure that no foreign materials or substances enter the product stream. The development of co-rotating twin screw extruders with sanitary design features has enabled manufacturers to produce food grade masterbatches that meet both regulatory requirements and performance specifications.
Wanplas Group, through its partnership with Nanjing Kerke Extrusion Equipment Company, provides advanced co-rotating twin screw extruders specifically designed for food grade masterbatch production. The Kerke KTE Series twin screw extruders incorporate comprehensive sanitary design features including stainless steel construction, polished surfaces, clean-out capabilities, and compliance with food contact regulations. These machines are particularly well-suited for food grade masterbatch production where regulatory compliance, cleanability, and consistent mixing quality are essential for achieving product specifications.
Formulation Ratios (Different Types)
The formulation of food grade masterbatches varies significantly depending on the required color, functional properties, food contact regulations, and end-use applications. Food grade masterbatches typically fall into several categories based on the type of colorant or additive used, including titanium dioxide-based white masterbatches, organic colorant masterbatches, nucleating agent masterbatches, and antistatic masterbatches for food packaging. Each category requires different formulation approaches and regulatory considerations to achieve optimal performance while maintaining compliance with food contact regulations.
Titanium dioxide-based white masterbatches represent one of the most common food grade formulations, utilizing food-grade titanium dioxide approved for direct food contact according to FDA 21 CFR 178.32970 or EU Regulation No 10/2011. A typical formulation for food grade titanium dioxide white masterbatch consists of 50% to 70% food-grade titanium dioxide, 25% to 45% carrier polymer, and 5% to 10% processing aid and dispersing agent. The titanium dioxide loading directly affects the opacity and tinting strength of the final product. For applications requiring high opacity, titanium dioxide loading typically ranges from 60% to 70%. For applications where tinting strength is more important than opacity, loading typically ranges from 50% to 60%. The choice of carrier polymer depends on the food contact compliance and compatibility with the final application matrix, with polyethylene, polypropylene, and polyester being common carrier polymers for food applications.
Organic colorant masterbatches use FDA-approved organic pigments to achieve specific colors in food packaging applications. These masterbatches typically contain 20% to 40% organic pigment, 55% to 75% carrier polymer, and 5% to 10% processing aid and dispersing agent. Organic pigments typically have higher tinting strength than inorganic pigments, allowing lower loadings while achieving strong coloration. A typical formulation for food grade red color masterbatch might include 30% organic red pigment approved for food contact, 65% polypropylene carrier, and 5% dispersing agent. For yellow color masterbatch, a typical formulation might include 25% organic yellow pigment, 70% polypropylene carrier, and 5% dispersing agent. The exact pigment loading must be optimized based on the specific pigment tinting strength and the required color intensity in the final application. All organic pigments must be approved for food contact according to applicable regulations, and migration limits must be considered in formulation design.
Nucleating agent masterbatches are used to control crystallinity and improve transparency in food packaging applications. These masterbatches typically contain 5% to 15% nucleating agent, 80% to 90% carrier polymer, and 5% to 10% processing aid. Nucleating agents for food applications must be approved for food contact and typically include sodium benzoate or specific nucleating agents approved for use in polyolefins. A typical formulation for food grade nucleating agent masterbatch might include 10% sodium benzoate nucleating agent, 85% polypropylene carrier, and 5% processing aid. The nucleating agent loading affects the degree of crystallinity control and the balance between transparency and mechanical properties. Higher loadings provide greater nucleating effect but may affect other properties if not properly balanced.
Antistatic masterbatches for food packaging use food-grade antistatic agents to dissipate static charges while maintaining compliance with food contact regulations. These masterbatches typically contain 15% to 30% migratory antistatic agent approved for food contact, 65% to 80% carrier polymer, and 5% to 10% processing aid. Food-grade antistatic agents must be approved for use in food contact materials and must not migrate at levels that could affect food safety. A typical formulation for food grade antistatic masterbatch might include 20% ethoxylated amine antistatic agent approved for food contact, 75% polyethylene carrier, and 5% processing aid. The migratory nature of these antistatic agents requires consideration of migration limits in formulation design, and the masterbatch loading in the final application must be controlled to ensure compliance with specific migration limits.
Combination masterbatches for food applications may include multiple functional additives to achieve multiple performance objectives. For example, color masterbatches may also include nucleating agents or processing aids to provide multiple functions in one masterbatch. A typical combination formulation for food packaging might include 25% organic pigment, 8% nucleating agent, 60% carrier polymer, and 7% dispersing agent and processing aid. These combination formulations must ensure that all components are approved for food contact and that the total migration from all components remains within regulatory limits. The interaction between different additives must be considered to ensure that additive stability is maintained during processing.
Production Process
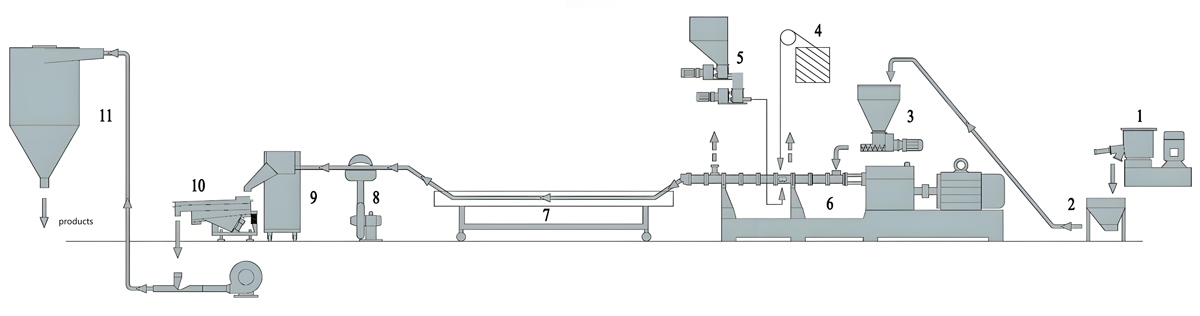
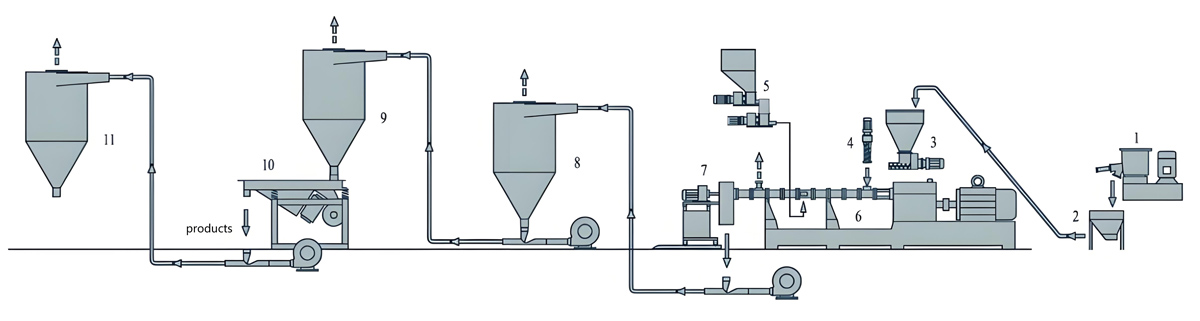
The production of food grade masterbatch using co-rotating twin screw extruders involves multiple carefully controlled stages that must be optimized to achieve consistent quality while maintaining regulatory compliance. The process begins with raw material preparation, progresses through compounding and pelletizing, and concludes with quality control and packaging. Each stage requires specific attention to cleanliness, traceability, and compliance with food contact regulations to ensure that the final product meets both quality and regulatory requirements.
Raw material preparation is a critical first step that significantly influences final product quality and regulatory compliance. All raw materials including carrier polymers, colorants, additives, and processing aids must be accompanied by documentation demonstrating compliance with food contact regulations. Materials should be stored in clean, dry conditions that prevent contamination from external sources. Carrier polymers should be verified to meet food contact regulations for the intended application, with documentation available for traceability purposes. Colorants and additives must be specifically approved for food contact according to applicable regulations, and supplier documentation must be retained. All raw materials must be weighed with high precision, typically to within 0.1% accuracy, to ensure consistent batch-to-batch composition. Weighing should be performed in clean areas using calibrated equipment to prevent contamination. All weighing records must be retained for traceability purposes and to demonstrate compliance with formulation specifications.
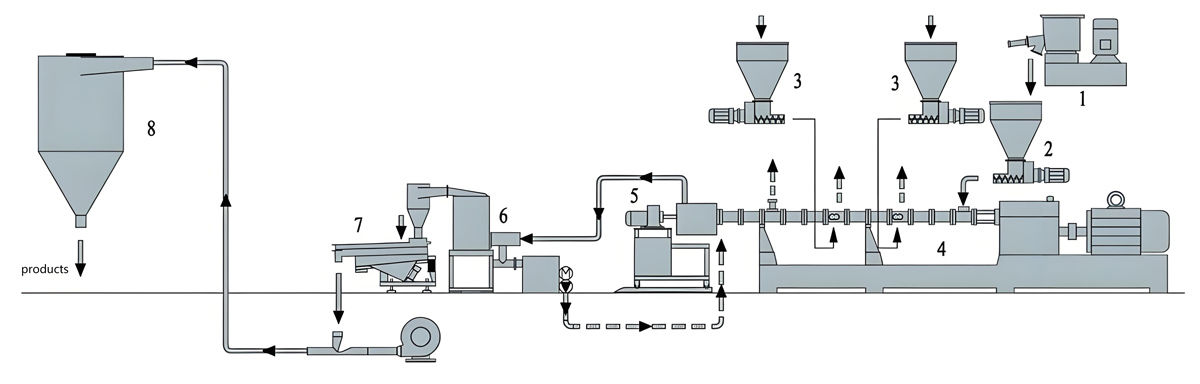
Feeding of raw materials into the co-rotating twin screw extruder requires precise metering and careful handling to maintain material cleanliness and prevent contamination. Food grade materials should be fed using feeding systems designed for clean operation, with enclosed feeders that prevent contamination from the environment. Gravimetric feeders should be used for accurate metering of all components, including carrier polymers, colorants, and additives. Feeder systems must be constructed from materials compatible with food contact, typically stainless steel or food-grade materials. The feed system should be designed for easy cleaning to prevent buildup of material between production runs. Feed zones should be equipped with temperature control to maintain optimal feeding conditions without causing material degradation. Food contact surfaces in the feed system must be smooth and polished to prevent material buildup and facilitate cleaning.
The compounding process in the co-rotating twin screw extruder occurs through a series of carefully designed barrel zones, each optimized for specific processing functions. The initial feed zone operates at lower temperatures, typically 160°C to 180°C for polyolefins, to ensure smooth feeding without premature melting. As material progresses through the extruder, temperature gradually increases, with the melting zone reaching 190°C to 210°C for optimal melting of polyolefin carriers. The high shear mixing zones, where dispersion of colorants and additives occurs, typically operate at peak processing temperatures of 200°C to 220°C. These temperatures must be carefully controlled to prevent thermal degradation while providing sufficient energy for dispersion. The vent zone typically operates at slightly lower temperatures of 190°C to 200°C to facilitate removal of any volatiles without causing excessive material degradation. The die zone typically operates at 210°C to 230°C to ensure proper flow through the die. All temperature setpoints must be compatible with the thermal stability of food contact materials used in the formulation.
The co-rotating design of the twin screws provides self-cleaning characteristics that are particularly beneficial for food grade production. The intermeshing screw configuration continuously wipes the screw surfaces, preventing material buildup that could cause contamination or degradation. This self-cleaning action helps maintain material purity and facilitates clean-out procedures between product changes. The screw geometry must be optimized for the specific formulation to achieve adequate dispersion while maintaining the self-cleaning characteristics. Screw configurations typically include conveying elements for material transport, kneading blocks for mixing and dispersion, and possibly reverse elements to enhance mixing. The screw design must balance mixing intensity with residence time to achieve optimal dispersion while preventing material degradation.
Melt filtration is an essential step in food grade masterbatch production, removing oversized particles, agglomerates, and contaminants that could affect product quality or regulatory compliance. Melt filters with mesh sizes ranging from 150 to 400 microns are commonly used, with the exact selection depending on the colorant particle size and dispersion quality requirements. Filter changers must be designed for sanitary operation with easy access for cleaning and filter changes. The filtration system must maintain temperature stability during filter changes to prevent material degradation. Screen changers with continuous operation capabilities are preferred for food grade production to minimize interruptions and maintain process consistency. All melt contact surfaces in the filtration system must be constructed from food-grade materials and designed for easy cleaning to prevent contamination between production runs.
Pelletizing of the compounded food grade masterbatch typically uses strand pelletizers or underwater pelletizers depending on the material characteristics and desired pellet shape. Strand pelletizing is common for polyolefin-based masterbatches due to the material’s tendency to form clean strands that cut cleanly. The pelletizing area must be maintained in clean conditions to prevent contamination. Pelletizer knives must be constructed from food-grade materials and kept sharp to ensure clean cuts that minimize fines generation. The pelletizing process must be controlled to achieve consistent pellet size, typically 2mm to 4mm in diameter and 3mm to 6mm in length, which ensures uniform feeding in downstream processing equipment. All pelletizing equipment must be designed for sanitary operation with easy access for cleaning and maintenance.
Cooling of the pellets after pelletizing is critical to prevent thermal degradation and maintain material properties. Air cooling systems with filtered air should be used to prevent contamination from the environment. Cooling air temperatures should be maintained between 15°C and 25°C to achieve proper cooling without causing thermal stress. The pellets must be cooled sufficiently to prevent sticking during subsequent handling, typically to below 50°C, but not overcooled to avoid thermal stress that could affect material properties. The cooling system must be designed to prevent contamination, with filters on air intakes and regular cleaning to prevent dust accumulation. Cooling conveyor systems should be constructed from food-grade materials and designed for easy cleaning.
Quality control procedures include measurement of color properties, dispersion quality assessment, migration testing, and regulatory compliance verification. Color measurement using spectrophotometers ensures that color development during processing remains within specified tolerances. Dispersion quality is assessed through microscopic examination of pellet cross-sections, looking for uniform distribution of colorants and additives without significant agglomeration. Migration testing must be performed to verify that the masterbatch complies with applicable migration limits for food contact materials. This testing typically involves extracting the masterbatch with appropriate food simulants and analyzing the extract for migrating substances. Additional quality tests may include melt flow index testing to ensure consistent processing characteristics and thermal analysis to verify material stability. All quality control records must be retained to demonstrate compliance with regulatory requirements and product specifications.
Production Equipment Introduction
The production of food grade masterbatch requires specialized co-rotating twin screw extruders capable of meeting regulatory requirements while maintaining mixing performance. Co-rotating twin screw extruders incorporate multiple advanced technologies to achieve the necessary sanitary design, cleanability, and compliance with food contact regulations. These machines are particularly important for food grade production where regulatory compliance and product purity are essential requirements.
Co-rotating twin screw extruders feature sanitary design characteristics that are essential for food grade production. All material contact surfaces are typically constructed from stainless steel with appropriate polish levels to facilitate cleaning and prevent material buildup. Stainless steel grades such as 304 or 316 are commonly used, with 316 being preferred for its superior corrosion resistance. Surface finishes on material contact areas typically meet sanitary requirements with surface roughness values below 0.8 microns Ra to ensure smooth, easily cleaned surfaces. All welds are ground smooth to prevent crevices where material could accumulate and potentially cause contamination. The design minimizes dead spaces and difficult-to-clean areas to ensure thorough cleaning between production runs.
Self-cleaning screw designs characteristic of co-rotating twin screw extruders are particularly beneficial for food grade production. The intermeshing screw configuration continuously wipes the screw surfaces, preventing material buildup and facilitating complete clean-out between product changes. This self-cleaning action helps maintain material purity and prevents cross-contamination between different product runs. Screw elements are typically designed with precise geometries to optimize the self-cleaning action while maintaining adequate mixing performance. The modular nature of many co-rotating screw designs allows easy reconfiguration for different formulations while maintaining the self-cleaning characteristics.
Easy clean-out and sanitation capabilities are essential features of food grade co-rotating extruders. The design should allow rapid disassembly and cleaning of material contact components. Quick-release mechanisms on die adapters, barrels, and other components facilitate access for cleaning and maintenance. All components should be designed to withstand frequent cleaning procedures without degradation. CIP (clean-in-place) capabilities may be incorporated to allow automated cleaning without complete disassembly. The design should facilitate drainage to prevent pooling of cleaning solutions that could cause contamination. All cleaning procedures must be documented to demonstrate consistent sanitation practices.
Traceability and documentation capabilities are essential for food grade production equipment. The extruder should be equipped with data logging systems that record all critical process parameters including temperatures, pressures, screw speeds, and feed rates. This data must be retained to demonstrate compliance with process specifications and to support traceability requirements. Batch tracking systems may be incorporated to allow tracking of specific production batches from raw material receipt through final product. The control system should provide easy export of production data for record-keeping purposes and regulatory compliance documentation.
Material contact surface compliance with food contact regulations is a fundamental requirement for food grade extruders. All materials in contact with the product must comply with applicable food contact regulations including FDA 21 CFR, EU Regulation No 10/2011, and other relevant standards. Material suppliers must provide documentation demonstrating compliance with these regulations. Regular inspection of material contact surfaces must be performed to verify that surfaces remain in good condition and have not degraded through use or cleaning procedures. Any replacement components must use materials that comply with the same food contact regulations as original equipment.
Kerke KTE Series twin screw extruders from Nanjing Kerke Extrusion Equipment Company represent advanced co-rotating solutions specifically designed for food grade masterbatch production. The KTE Series incorporates comprehensive sanitary design features including stainless steel construction, polished surfaces, easy clean-out capabilities, and compliance with food contact regulations. These extruders are available in screw diameters from 20mm to 120mm, with length-to-diameter ratios of 40:1 to 60:1 to provide sufficient mixing length for food grade additive dispersion. The KTE Series features modular construction that allows easy maintenance and reconfiguration for different masterbatch formulations, with quick-change barrel segments and screw elements that facilitate rapid product changeovers while maintaining sanitary design compliance.
Parameter Settings
Proper parameter settings are essential for achieving consistent quality in food grade masterbatch production using co-rotating twin screw extruders. Temperature profiles, screw speeds, feed rates, vent conditions, and other process parameters must be optimized for each specific formulation to achieve optimal dispersion while maintaining regulatory compliance. Parameter optimization requires consideration of food contact material properties, equipment capabilities, and regulatory requirements.
Temperature profiles must be carefully configured to ensure proper melting, dispersion, and devolatilization while preventing thermal degradation of food contact materials. For typical food grade masterbatch production using polyolefin carriers, the temperature profile might be: Feed zone 160°C to 170°C, melting zone 180°C to 200°C, mixing zone 200°C to 220°C, vent zone 190°C to 200°C, and die zone 210°C to 230°C. The exact temperatures must be adjusted based on the specific carrier polymer, colorant type, and required product properties. Higher colorant loadings may require slightly higher temperatures to achieve proper dispersion, though this must be balanced against increased thermal degradation risk. Temperature uniformity across the barrel cross-section is critical for consistent processing, requiring precise heater control algorithms and adequate cooling capacity. Temperature fluctuations must be maintained within tight tolerances, typically plus or minus 2°C of setpoint, to prevent thermal degradation and ensure consistent product quality.
Screw speed significantly affects dispersion quality, residence time, and the self-cleaning characteristics of co-rotating extruders. For food grade masterbatch production, screw speeds typically range from 150 to 350 rpm depending on screw diameter and material characteristics. Smaller extruders with 20mm to 40mm screw diameters typically operate at higher speeds of 250 to 350 rpm to achieve sufficient mixing intensity. Medium-sized extruders with 50mm to 80mm screw diameters typically operate at 200 to 300 rpm. Large extruders with 90mm to 120mm screw diameters typically operate at 150 to 250 rpm. The optimal screw speed balances mixing intensity with residence time, ensuring sufficient dispersion while maintaining the self-cleaning characteristics. Screw speed also affects residence time, which influences the extent of dispersion and the potential for thermal degradation. The self-cleaning action of co-rotating screws is maintained across a range of speeds, though optimal performance may be achieved at specific speed ranges depending on screw geometry.
Feed rates must be optimized to maintain consistent residence time and filling level in the extruder. Higher feed rates increase throughput but reduce residence time, potentially compromising dispersion quality. Lower feed rates improve dispersion but reduce productivity and may increase thermal degradation due to longer residence time. For food grade masterbatch with 20% to 40% additive loading, typical feed rates range from 50 to 200 kg per hour for 20mm to 40mm extruders, 200 to 800 kg per hour for 50mm to 80mm extruders, and 800 to 3000 kg per hour for 90mm to 120mm extruders. The feed rate should be adjusted to maintain a partially filled condition in the mixing zones, which allows sufficient time for dispersion without causing excessive residence time that could degrade thermal stability. Feed rates also affect the degree of fill in the extruder, which influences the self-cleaning action and dispersion quality.
Vent vacuum levels must be optimized to remove moisture, volatiles, and entrapped air from the melt. Inadequate venting can cause voids, bubbles, and surface defects in the pellets, while excessive venting can cause material loss and increased energy consumption. Typical vent vacuum levels range from 700 to 900 mbar absolute pressure for food grade masterbatch production. The vacuum level should be adjusted based on the moisture content of raw materials and the volatility of processing aids. Materials with higher moisture content require stronger vacuum to achieve effective drying, while volatile processing aids may require reduced vacuum to prevent excessive material loss through the vent. Vent systems must be designed for sanitary operation with easy access for cleaning to prevent contamination between production runs.
Die temperature and pressure settings affect pellet quality and consistency. Die temperatures are typically set 5°C to 10°C above the final barrel zone temperature to ensure proper flow through the die. For food grade masterbatch, die temperatures typically range from 215°C to 240°C. Die pressure typically ranges from 50 to 150 bar depending on material viscosity and throughput rate. Higher die pressures can improve pellet definition but increase equipment stress. The optimal die pressure balances pellet quality with equipment longevity. Die pressure monitoring systems should be installed with high-pressure alarms and automatic shutdown capabilities to prevent overpressure events that could compromise material purity. Die lip temperature must be controlled precisely to prevent material hang-up that could degrade and potentially cause contamination.
Equipment Price
The investment required for co-rotating twin screw extruder systems for food grade masterbatch production varies significantly based on equipment size, configuration, sanitary design features, and regulatory compliance requirements. Understanding the cost structure and pricing factors helps manufacturers make informed investment decisions and budget appropriately for equipment acquisition. Prices are typically quoted in US dollars for international transactions.
Co-rotating twin screw extruders with sanitary design features are available in various size categories with corresponding price ranges. Small laboratory or pilot-scale extruders with 20mm to 25mm screw diameters typically range from USD 55,000 to USD 85,000 depending on sanitary design features and configuration. These small extruders typically have throughput capacities of 10 to 50 kg per hour and are suitable for formulation development and small-scale production with appropriate sanitary features. Medium-sized production extruders with 40mm to 60mm screw diameters typically range from USD 150,000 to USD 300,000 depending on specifications and sanitary design level. These extruders typically achieve throughput rates of 100 to 500 kg per hour and represent the most common size range for food grade masterbatch production. Large production extruders with 80mm to 120mm screw diameters typically range from USD 400,000 to USD 900,000 or more depending on configuration and sanitary design requirements. These large extruders can achieve throughput rates of 800 to 3000 kg per hour and are suitable for high-volume production facilities.
Sanitary design features represent a significant cost component, typically adding 25% to 40% to the base extruder price compared to standard extruders without sanitary features. Stainless steel construction with appropriate surface polish typically costs USD 15,000 to USD 45,000 more than standard construction depending on extruder size and the grade of stainless steel used. Sanitary design features including polished surfaces, easy clean-out capabilities, and minimized dead spaces typically add USD 12,000 to USD 35,000. Documentation and compliance verification for food contact regulations typically add USD 5,000 to USD 15,000 depending on the extent of certification required. Data logging and traceability systems for regulatory compliance typically cost USD 8,000 to USD 20,000 depending on the sophistication of the system. The total premium for sanitary design and compliance features typically ranges from USD 40,000 to USD 115,000 depending on extruder size and the extent of sanitary features specified.
Kerke KTE Series twin screw extruders from Nanjing Kerke Extrusion Equipment Company offer competitive pricing in the market while providing comprehensive sanitary design capabilities. For KTE Series extruders with sanitary design, typical pricing includes: KTE-25 (25mm screw diameter) approximately USD 60,000 to USD 85,000 with basic sanitary features, KTE-40 (40mm screw diameter) approximately USD 160,000 to USD 220,000 with standard sanitary design, KTE-60 (60mm screw diameter) approximately USD 240,000 to USD 320,000 with comprehensive sanitary features, KTE-80 (80mm screw diameter) approximately USD 420,000 to USD 580,000 with full sanitary design including traceability systems, and KTE-120 (120mm screw diameter) approximately USD 650,000 to USD 900,000 with complete sanitary design including advanced compliance documentation. These prices typically include the extruder with basic sanitary features, standard control system with data logging, and basic accessories. Custom configurations, additional accessories, and advanced sanitary features will increase the final price.
Feeding systems represent a significant additional cost for food grade masterbatch production. Gravimetric feeding systems with sanitary design typically cost USD 15,000 to USD 40,000 per feeder depending on accuracy requirements, material handling capacity, and sanitary design level. Food grade masterbatch production typically requires at least two gravimetric feeders, one for the carrier polymer and one for the colorant or additive, with additional feeders for additives if required. Feeder systems should be constructed from food-grade materials with easy clean-out capabilities. Liquid dosing systems for processing aids typically cost USD 10,000 to USD 25,000 per dosing system with sanitary design features. Bulk material handling systems for food grade applications can cost an additional USD 35,000 to USD 100,000 depending on capacity and sanitary design level.
Pelletizing systems represent another significant cost component with sanitary requirements. Strand pelletizing systems typically cost USD 25,000 to USD 65,000 depending on throughput capacity and automation level, with additional costs for sanitary design of USD 12,000 to USD 30,000. Underwater pelletizing systems typically cost USD 40,000 to USD 90,000 depending on capacity, with sanitary design adding USD 18,000 to USD 40,000. Cooling systems for strand pelletizing typically cost USD 15,000 to USD 30,000 depending on capacity and sanitary design requirements. Complete pelletizing packages including cutting, cooling, conveying, and sanitary design typically range from USD 50,000 to USD 150,000 depending on throughput and automation.
Complete turnkey production lines including extruder, sanitary feeding systems, pelletizing, material handling, and control systems typically cost: Small pilot-scale lines with 20mm to 25mm extruders approximately USD 150,000 to USD 250,000, medium-scale production lines with 40mm to 60mm extruders approximately USD 350,000 to USD 800,000, and large-scale production lines with 80mm to 120mm extruders approximately USD 800,000 to USD 2,200,000 or more. These complete line prices include all major equipment, sanitary design integration, compliance documentation, startup support, and basic training. Additional costs for facility preparation including sanitation facilities, documentation systems, and operator training are not included in equipment prices and should be budgeted separately, typically adding 20% to 35% to the equipment investment depending on facility conditions and regulatory requirements.
Production Problems and Solutions
Despite careful process optimization and sanitary design, food grade masterbatch production can encounter various problems that affect product quality, regulatory compliance, or operational efficiency. Understanding common problems, their causes, and implementing effective solutions is essential for maintaining consistent production and regulatory compliance. Each problem requires specific diagnostic approaches and corrective actions to address root causes while maintaining sanitary conditions.
Color inconsistency in the finished masterbatch represents one of the most common quality problems. This problem can manifest as color variations between batches, inconsistent tinting strength, or failure to meet specified color requirements. The most common cause is inconsistent colorant loading in the formulation, which can occur from weighing errors during raw material preparation, segregation during feeding, or colorant degradation during processing. Another frequent cause is poor dispersion of colorants resulting in non-uniform color distribution and inconsistent appearance. Inadequate dispersion can result from insufficient mixing intensity, improper screw configuration, or temperature profiles that cause colorant degradation or reagglomeration. Inconsistent raw material quality between colorant batches can also cause color variations. Thermal degradation of colorants during processing can cause color changes and reduce tinting strength.
Addressing color inconsistency requires systematic investigation of multiple potential causes. The first step is verification of the formulation by checking weighing records for raw material preparation and confirming that actual ingredient quantities match the target formulation. If formulation errors are identified, the batch should be recompounded with corrected ingredient quantities. For segregation problems, the feeding system should be inspected and modified to ensure uniform mixing of colorants with carrier polymer, possibly through the use of pre-mixing systems or improved feeder design. For dispersion problems, screw configuration should be reviewed to ensure sufficient mixing elements are present, and screw speed may be increased to enhance dispersion intensity. Temperature profiles should be reviewed to ensure that temperatures are optimized for dispersion without causing thermal degradation. If thermal degradation of colorants is suspected, residence time may be reduced by adjusting feed rate or screw speed, or processing temperatures may be reduced if thermal stability is a concern. Material quality should be verified through testing of incoming colorants to ensure consistent tinting strength and color characteristics.
Preventing color inconsistency problems requires implementation of preventive measures during formulation development and ongoing quality control. Formulation development should include margin in colorant loading to account for normal variations in raw material properties and processing conditions. Process validation should establish acceptable processing windows for temperature, screw speed, and residence time that prevent colorant degradation while ensuring adequate dispersion. Regular quality control testing should include color measurement on each production batch using standardized spectrophotometric methods to detect developing problems before they affect customer deliveries. Raw material specifications should include color requirements that ensure incoming colorants meet quality standards. Equipment maintenance programs should include regular inspection of mixing elements to ensure they are not worn, which can reduce mixing effectiveness. Process monitoring should include in-line color measurement if available, providing real-time feedback on color consistency.
Contamination from previous production runs represents a serious compliance problem in food grade masterbatch production. Cross-contamination can occur from inadequate cleaning between product changes, material buildup in equipment, or foreign materials entering the process stream. Contamination can affect product quality and potentially cause regulatory compliance issues if non-food-grade materials contaminate food grade products. The primary causes include inadequate cleaning procedures between production runs, material buildup in areas that are difficult to clean, or improper material handling procedures that allow foreign materials to enter the process. The self-cleaning characteristics of co-rotating screws help reduce but do not eliminate contamination risks.
Addressing contamination problems requires thorough cleaning and process adjustment. The first step is identification of the contamination source through analysis of contaminated material and inspection of equipment. Complete cleaning of all material contact surfaces is necessary, potentially including disassembly of equipment for detailed cleaning. Cleaning procedures should be reviewed and potentially modified to ensure more effective cleaning. Equipment design may need modification to eliminate difficult-to-clean areas where material accumulates. Material handling procedures should be reviewed and reinforced to prevent foreign materials from entering the process. If contamination is from previous production runs, the cleaning procedures between product changes must be made more rigorous and potentially extended to ensure complete removal of previous materials. The entire production system should be sanitized after any contamination event before resuming production of food grade materials.
Preventing contamination problems requires implementation of comprehensive sanitation programs and equipment design considerations. Equipment should be designed with sanitary features including smooth surfaces, minimal dead spaces, and easy access for cleaning. Cleaning procedures should be standardized and documented, with verification of cleaning effectiveness through visual inspection and potentially testing for residual materials. Regular deep cleaning procedures should be implemented beyond routine cleaning to prevent gradual buildup in difficult areas. Material handling procedures should emphasize clean operation to prevent contamination from the environment or handling equipment. Personnel training should emphasize the importance of maintaining clean conditions and following proper procedures. Regular audits of sanitation practices should be performed to ensure consistent implementation of cleaning procedures and identify areas for improvement.
Migration test failures represent a serious regulatory compliance problem in food grade masterbatch production. Migration occurs when substances from the masterbatch migrate into food or food simulants at levels exceeding regulatory limits. Failures can result from use of non-compliant additives, additive concentrations exceeding migration limits, degradation products forming during processing, or contamination from non-food-grade sources. Migration failures can require product rejection and potentially product recalls, representing significant financial and regulatory consequences.
Addressing migration test failures requires systematic investigation of potential sources of migrating substances. The first step is analysis of the failed migration test to identify the specific substances that exceeded limits. Formulation records must be reviewed to verify that all components are approved for food contact at the used concentrations. If non-compliant materials are identified, they must be replaced with compliant alternatives and the formulation revalidated. If additive concentrations are causing migration failures, concentrations may need to be reduced or alternative additives with lower migration potential should be identified. If degradation products are identified as migrating substances, processing conditions may need to be modified to reduce thermal degradation, possibly by reducing temperatures or residence time. If contamination is identified as the source, cleaning procedures must be reviewed and potentially modified.
Preventing migration test failures requires comprehensive formulation development and process optimization. Formulation development must include migration testing as part of the validation process for new formulations or process changes. All additives must be verified for compliance with applicable food contact regulations at the intended use levels. Process optimization should minimize thermal degradation and formation of degradation products. Migration testing should be performed regularly as part of quality control to detect developing problems before they affect customer deliveries. Documentation of all material approvals and migration testing results must be maintained to demonstrate compliance with regulatory requirements. Regular review of regulatory requirements is necessary to ensure that formulations remain compliant as regulations evolve.
Additive dispersion problems resulting in agglomeration of colorants or additives represent a quality issue that can cause inconsistent appearance and potentially affect migration characteristics. Agglomerates appear as visible specks or inclusions in molded parts or extruded products and can cause color variations. The primary causes of agglomeration include insufficient dispersing agent concentration, inadequate mixing intensity, excessive screw speed causing additive degradation and reagglomeration, temperature profiles that cause dispersing agents to volatilize prematurely, or poor initial dispersion due to inadequate wetting of additives by the carrier polymer. High additive loadings increase the difficulty of achieving uniform dispersion and make agglomeration more likely if processing conditions are not optimized.
Solving agglomeration problems requires addressing the underlying dispersion issues through formulation and process adjustments. The first approach is to increase dispersing agent concentration in the formulation, typically by 2% to 5% depending on the severity of the problem. Different dispersing agents with improved wetting characteristics for the specific additive may be tested, including polymeric dispersants or surfactant-type dispersants with specific affinity for additive surfaces. Screw configuration should be reviewed to ensure sufficient mixing elements are present, and screw speed may be adjusted to optimize shear conditions. Temperature profiles should be adjusted to ensure that dispersing agents remain active throughout the mixing zones, which may require lower temperatures in early zones and higher temperatures in mixing zones to optimize dispersion efficiency. Pre-mixing of additives with carrier polymer before feeding into the extruder can improve initial dispersion and reduce the likelihood of agglomeration.
Preventing agglomeration problems requires attention to multiple factors throughout the production process. Raw material quality control should include particle size analysis of additives to ensure they meet specifications, as overly coarse additives are more difficult to disperse. Dispersing agent selection should consider compatibility with both the additive and carrier polymer, with testing performed on multiple candidates to identify the most effective options. Process development should include optimization of screw configuration and processing parameters specifically for dispersion quality, potentially using advanced mixing elements or multiple mixing zones. Regular quality control should include microscopic examination of pellet cross-sections to detect developing dispersion problems before they cause customer complaints. Equipment maintenance programs should include regular inspection of mixing elements to ensure they are not worn, which can reduce mixing effectiveness.
Cross-contamination between different food grade products represents a compliance and quality problem that can affect traceability and regulatory compliance. Cross-contamination can occur when material from one product remains in equipment during production of a different product, potentially causing allergen issues or affecting product specifications. The primary causes include inadequate cleaning between product changes, material buildup in dead spaces, or incomplete purging of the extruder system between products. Even small amounts of cross-contamination can be problematic if the products have different specifications or if allergens are present.
Addressing cross-contamination requires thorough cleaning and purging procedures. The first step is identification of the extent of cross-contamination through product testing. Complete cleaning and purging procedures must be implemented, potentially including disassembly of equipment for detailed cleaning. Cleaning procedures may need to be extended beyond standard procedures to ensure complete removal of all previous product material. Purging compounds may be used to assist in removal of previous materials, particularly if the materials have different characteristics. After cleaning and purging, verification through product testing should be performed to confirm that cross-contamination has been eliminated before resuming production of the next product. Documentation of cleaning procedures and verification results must be maintained for traceability purposes.
Preventing cross-contamination requires implementation of robust cleaning and purging protocols specifically designed for product changeovers. Equipment should be designed with easy clean-out capabilities to facilitate thorough cleaning between products. Standardized cleaning procedures should be developed for different types of product changes, with more extensive procedures for products with different characteristics or where allergens are present. Purging procedures should be optimized for effectiveness while minimizing material waste. Verification testing after each product changeover should be performed to confirm that cross-contamination has been eliminated. Production planning should consider product compatibility and sequence changes to minimize the risk of cross-contamination, possibly grouping similar products together in production schedules.
Maintenance
Regular maintenance is essential for maintaining optimal performance and regulatory compliance in food grade masterbatch production. The sanitary design requirements, the need for consistent product quality, and regulatory traceability requirements all contribute to the importance of comprehensive maintenance programs. Implementing preventive and predictive maintenance programs helps minimize unplanned downtime, maintain product quality, ensure regulatory compliance, and reduce total cost of ownership over the equipment lifespan.
Daily maintenance tasks focus on inspection and minor adjustments that ensure reliable operation during the production shift. At the start of each shift, operators should perform visual inspections of the extruder and auxiliary equipment to identify obvious problems such as leaks, loose components, or abnormal noises. Temperature readings from all barrel zones should be recorded and compared to normal operating ranges to identify developing temperature control problems. Pressure readings from the melt pressure transducer should be monitored to ensure consistent die pressure. The feeding system should be inspected for proper material flow and accurate metering. All material contact surfaces should be visually inspected for signs of material buildup or contamination that could affect product quality or compliance.
Weekly maintenance tasks involve more detailed inspections and preventive maintenance activities. Screw and barrel inspection through the hopper should be performed to look for signs of excessive wear, surface degradation, or material buildup that could affect performance or contamination risk. The vent system should be inspected and cleaned if necessary to prevent accumulation of material that could reduce venting effectiveness or cause contamination. The die assembly should be disassembled for inspection of wear patterns and cleaning of flow surfaces, with attention to removing any material deposits that could affect product quality. Pelletizer knives should be inspected for sharpness and proper alignment, with resharpening or replacement as needed. Control system calibration checks should be performed on temperature controllers, pressure transducers, and feed rate indicators to ensure accurate control. All material contact surfaces should be inspected for signs of wear, corrosion, or degradation that could affect sanitation or compliance.
Monthly maintenance tasks focus on more extensive preventive maintenance and condition monitoring. The drive system including electric motor, gearbox, and couplings should be inspected for signs of wear, overheating, or abnormal vibration. Motor current readings should be recorded and trended to identify developing problems. Gearbox oil should be sampled and analyzed for wear particles, with oil replacement scheduled based on analysis results or at least annually regardless of analysis results. Bearing temperatures should be monitored for elevations that could indicate lubrication problems or wear. Screw and barrel wear should be measured using appropriate gauges, with measurements recorded to track wear rates over time. Feed system calibration checks should be performed using standard weights and flow rates to verify accuracy. The complete extruder should be cleaned thoroughly to remove any material buildup that could affect sanitation or compliance.
Semi-annual maintenance tasks involve major component inspection and replacement based on condition monitoring results and maintenance schedules. Screw elements should be removed for detailed inspection and measurement of wear dimensions. Elements with wear exceeding specified tolerances should be replaced to maintain mixing effectiveness and prevent material contamination. The barrel bore should be inspected for wear patterns, ovality, or damage, with replacement scheduled if wear affects sanitary properties or product quality. Heating elements should be inspected for proper operation and replaced if they show signs of degradation. Temperature sensors should be calibrated or replaced if they show drift from specified accuracy. The die and adapter should be removed for thorough cleaning and inspection of flow surfaces, with replacement if flow surfaces are significantly worn or damaged. All bearings in the drive system should be replaced preventively based on operating hours or condition monitoring results. All seals and gaskets should be replaced to prevent leakage and maintain sanitary properties.
Annual maintenance tasks involve comprehensive equipment assessment and major overhauls as required. The complete extruder should be disassembled for thorough inspection and measurement of all components. Wear measurements should be compared to previous measurements to track wear rates and identify components approaching end of life. Based on wear measurements and operating hours, a replacement schedule should be established for major components including screw, barrel, die, heating elements, and drive system components. The control system should be thoroughly tested and calibrated, with verification of safety interlocks, emergency shutdown systems, and data logging functionality. The complete feeding system should be disassembled, cleaned, inspected, and recalibrated. All material contact surfaces should be inspected for compliance with sanitary requirements, with refinishing or replacement if surfaces have degraded. Documentation should be reviewed and updated to ensure all maintenance activities are properly recorded and compliance documentation is current.
Predictive maintenance technologies can significantly improve maintenance effectiveness and reduce unplanned downtime for food grade masterbatch production equipment. Vibration monitoring of the drive system can detect bearing or gearbox problems before they cause catastrophic failure. Regular microscopic examination of product can detect changes in dispersion quality that may indicate screw wear. Temperature data logging and trend analysis can identify subtle changes in temperature control that may indicate heater degradation or sensor drift. Regular migration testing can detect changes in product composition that may indicate material degradation or contamination. These predictive maintenance techniques, combined with preventive maintenance schedules, enable more efficient maintenance planning and reduce the likelihood of unexpected equipment failures or compliance issues.
FAQ
Q: What food contact regulations apply to food grade masterbatch production?
A: Applicable regulations depend on the target market, but commonly include FDA 21 CFR for the United States, EU Regulation No 10/2011 for Europe, and various other national and international regulations. All materials in contact with food must comply with these regulations, and migration limits must be met. Kerke KTE Series extruders are designed to facilitate compliance with these regulations through sanitary design and documentation capabilities.
Q: How often should food grade masterbatch extruders be cleaned between production runs?
A: Cleaning frequency depends on the specific products being produced and the potential for cross-contamination. Product changeovers typically require cleaning between different products, particularly if the products have different colorants, additives, or allergen profiles. Routine cleaning should be performed at least weekly even when not changing products to prevent gradual material buildup. More extensive cleaning should be performed monthly or quarterly depending on production intensity and material types.
Q: What is the typical energy consumption for food grade masterbatch production?
A: Energy consumption depends on extruder size, throughput rate, material viscosity, and operating parameters. Typical specific energy consumption ranges from 0.2 to 0.4 kWh per kilogram of product for small extruders, and 0.15 to 0.3 kWh per kilogram for larger extruders. Higher additive loadings typically increase energy consumption due to higher melt viscosity. Energy consumption can be optimized by proper screw design, temperature profile optimization, and use of efficient drive systems.
Q: Can food grade masterbatch be produced on conventional twin screw extruders without sanitary design features?
A: While food grade masterbatch can technically be produced on conventional extruders, the lack of sanitary design features creates significant challenges in achieving and maintaining regulatory compliance. Conventional extruders may have difficult-to-clean areas, material buildup potential, and materials that may not comply with food contact regulations. Production on conventional extruders typically requires extensive cleaning procedures and may not meet all regulatory requirements without modifications. For consistent compliance and efficient production, sanitary extruders such as the Kerke KTE Series are strongly recommended.
Q: What is the maximum additive loading that can be processed in twin screw extruders for food grade applications?
A: The maximum practical additive loading depends on the additive type, particle characteristics, carrier polymer, and screw design. For titanium dioxide-based white masterbatches, practical maximum loading ranges from 60% to 75%. For organic colorant masterbatches, maximum loading is typically lower, ranging from 35% to 45% due to different tinting strength characteristics. Loadings above these ranges typically cause excessive viscosity, poor dispersion, and accelerated equipment wear. The optimal loading balances performance requirements with processability and regulatory compliance.
Q: How can migration compliance be ensured in food grade masterbatch production?
A: Migration compliance can be ensured by using only materials approved for food contact at the intended use levels, conducting migration testing as part of formulation validation, maintaining consistent processing conditions to prevent formation of degradation products, implementing rigorous cleaning procedures to prevent contamination, and regularly testing production batches for migration compliance. Documentation of all material approvals and test results is essential for demonstrating regulatory compliance.
Q: What are the key features to look for when selecting food grade masterbatch extruders?
A: Key features include stainless steel construction with appropriate surface polish for sanitation, easy clean-out design to prevent contamination, self-cleaning screw characteristics to minimize material buildup, documentation and traceability systems for regulatory compliance, data logging capabilities for process records, and verification of material compliance with food contact regulations. Kerke KTE Series extruders incorporate these essential features for food grade production.
Conclusion
The production of high-quality food grade masterbatches requires specialized co-rotating twin screw extruders, carefully optimized formulations, and rigorous attention to regulatory compliance and sanitation. Co-rotating twin screw extruders such as the Kerke KTE Series provide the necessary combination of mixing performance, self-cleaning characteristics, and sanitary design required for successful food grade masterbatch production. The selection of appropriate equipment, optimization of processing parameters, and implementation of comprehensive maintenance programs are all essential for achieving consistent product quality and regulatory compliance.
Formulation development must balance color and functional requirements with regulatory compliance considerations. All materials used in food grade masterbatches must comply with applicable food contact regulations, and migration limits must be carefully considered in formulation design. Proper selection of colorants, additives, and carrier polymers is essential for achieving desired performance while maintaining compliance. The complexity of regulatory requirements necessitates thorough documentation and traceability throughout the production process.
Production process optimization requires careful attention to temperature profiles, screw speeds, feed rates, and clean-out procedures to achieve optimal dispersion while maintaining regulatory compliance. The self-cleaning characteristics of co-rotating screws provide advantages in maintaining material purity and facilitating clean-out between production runs. Regular quality control testing is essential for monitoring process consistency and detecting developing problems before they affect product quality or regulatory compliance. Migration testing must be performed regularly to verify ongoing compliance with regulatory requirements.
Sanitation and compliance programs must address the regulatory requirements and cleanability needs of food grade production. Equipment must be designed with sanitary features that facilitate thorough cleaning and prevent material buildup. Regular cleaning procedures must be standardized and documented to ensure consistent implementation. Documentation of all material approvals, process parameters, and quality test results is essential for demonstrating regulatory compliance. Personnel training must emphasize the importance of maintaining clean conditions and following proper procedures.
Maintenance programs must address the specific requirements of food grade production, including the need to maintain sanitary surfaces and prevent contamination. Regular inspection and maintenance of material contact surfaces ensures they remain in compliance with sanitary requirements. Component inspection and replacement based on wear measurements helps optimize the balance between component cost and production impact. Predictive maintenance technologies can significantly improve maintenance effectiveness by enabling early detection of developing problems before they cause unplanned downtime or compliance issues.
For manufacturers seeking to establish or expand food grade masterbatch production capabilities, co-rotating twin screw extruders offer the necessary combination of performance, sanitation, and regulatory compliance for modern production facilities. The Kerke KTE Series from Nanjing Kerke Extrusion Equipment Company provides advanced sanitary design technology specifically designed for food grade masterbatch applications, offering competitive pricing and proven reliability. By implementing appropriate equipment, optimized processes, comprehensive sanitation programs, and rigorous quality control procedures, manufacturers can achieve consistent production of high-quality food grade masterbatches meeting the demanding requirements of food packaging, beverage containers, and other food contact applications while maintaining full compliance with regulatory requirements.