Introduction
Low migration masterbatch represents a critical segment of the plastic coloration industry, particularly for applications involving direct food contact, medical devices, and consumer goods subject to strict regulatory requirements. These masterbatches are formulated using additives and pigments that resist migration or diffusion from the polymer matrix to the surface or into the surrounding environment. The continuous twin screw extruder has emerged as the preferred equipment for low migration masterbatch production due to its precise process control, consistent mixing, and ability to handle formulation complexity.
Regulatory standards including FDA 21 CFR, EU 10/2011, and other regional regulations set strict limits on specific migration levels for plastic materials in food contact applications. Compliance requires specialized formulation design, careful raw material selection, and optimized processing techniques to minimize additive and pigment migration risks. The continuous twin screw extruder provides the controlled processing environment necessary to achieve low migration performance while maintaining color uniformity and mechanical properties.
This comprehensive guide explores the unique requirements of low migration masterbatch manufacturing, from formulation principles to processing optimization and quality assurance protocols. It addresses the technical challenges of producing masterbatch that meet strict regulatory standards while maintaining production efficiency and cost effectiveness. Whether establishing new production lines or optimizing existing operations, this guide provides essential insights for successfully navigating the complex landscape of low migration masterbatch production.
Key topics covered include formulation design principles, continuous extrusion technology specifics, process parameter optimization, migration testing methodologies, equipment selection criteria, and troubleshooting strategies. Special emphasis is placed on practical implementation of regulatory requirements throughout the production process. By understanding the critical factors affecting migration and the technology available to control it, manufacturers can produce high-quality low migration masterbatch that meets stringent industry standards.
Formulation Proportions (Different Types)
The formulation of low migration masterbatch differs significantly from standard masterbatch, requiring careful selection of materials and precise control of component ratios. Migration resistance depends on chemical compatibility between additive and polymer matrix, molecular weight of additives, and distribution uniformity throughout the polymer.
Food Contact Application Formulations
For food contact applications, formulation must comply with FDA indirect food additives regulations (21 CFR) and EU 10/2011. The formulation typically includes 2-15% pigment, 85-97% food-grade carrier polymer, and 0.5-2% dispersant that meets food contact requirements.
For a typical food contact white masterbatch, formulation consists of 10% FDA-compliant titanium dioxide (R104 or equivalent), 88.5% food-grade LDPE carrier, and 1.5% low molecular weight polyethylene wax approved for food contact. The carrier polymer must meet FDA 21 CFR 177.1520 for olefin polymers, specifying approved polymer types and additive limits.
Food contact color masterbatch formulations for use in packaging typically use organic pigments approved under 21 CFR 178.3297. Pigment loading varies based on pigment type and desired opacity. Phthalocyanine blue and green can achieve 10-15% loading, while azo reds and yellows may be limited to 5-10% due to specific migration limits.
Antioxidants and stabilizers must be selected from FDA-approved lists such as 21 CFR 178.2010. Typical antioxidants for polyolefin carriers include hindered phenols and phosphite compounds at 0.1-0.5% levels to provide processing stability without migration concerns.
Medical Device Application Formulations
Medical device masterbatch formulation must comply with ISO 10993 and USP Class VI biocompatibility requirements. The formulation is more restrictive, often requiring higher purity materials and tighter additive limits.
A typical medical device color masterbatch formulation consists of 5-10% medical grade pigment, 90-94% medical grade carrier polymer, and 1% non-migrating dispersant. Medical grade pigments must meet ISO 10993 biocompatibility standards and should not contain leachable heavy metals or residual solvents above specified limits.
Carrier polymers for medical applications are typically specialized grades of polyethylene or polypropylene with low residual monomer content and specified purity levels. For implantable device components, specialized implantable grade polymers may be required, and pigment loading is often limited to 5-7% to maintain mechanical properties.
Additive selection is extremely critical for medical device masterbatch. Only materials listed in the FDA Master File (DMF) or otherwise approved for specific medical applications should be used. Low migration dispersants including medical grade glycerol esters or specialized polyolefin waxes are required to avoid leaching concerns.
Baby Product Application Formulations
Baby product masterbatch requires compliance with specific safety standards including ASTM F963, EN 71, and regional regulations addressing infant product safety. These formulations emphasize non-toxic components and low migration rates to ensure product safety for sensitive users.
Formulation typically includes 5-10% non-toxic pigment, 89-94% virgin carrier polymer free from recycled content, and 1-2% dispersant approved for baby product use. Pigments must meet heavy metal limits specified in ASTM F963 Section 8, particularly for lead and cadmium content.
Colorants for baby products often use organic pigments or mineral oxides that meet stringent purity requirements. Inorganic pigments are preferred for durability and non-toxicity. Specialized water-based pigments may be used for specific applications requiring extreme purity.
Stabilizer selection is limited to materials with low potential for contact sensitization. Traditional hindered phenol antioxidants may be replaced with naturally derived alternatives where appropriate. All formulation components must be free from phthalates, bisphenol A, and other suspect chemical compounds.
High Temperature Application Formulations
High temperature applications such as microwaveable food containers and ovenware require masterbatch that maintains color and physical properties at elevated temperatures without migration. These formulations demand heat-resistant pigments and high-performance stabilizers.
Formulation for microwave-safe masterbatch typically includes 5-10% heat-resistant pigment, 88-94% high melting point carrier polymer, and 1-2% high-temperature dispersant. Carrier polymers such as high-density polyethylene or polypropylene with high melt flow properties ensure compatibility with microwave processing.
Heat-resistant pigments including quinacridone reds, phthalocyanine greens, and titanium dioxide with stable crystal structures are selected for their ability to withstand temperatures up to 250°C during processing and application. Specialized heat stabilizers may be added at 0.5-1.0% levels to maintain color integrity during thermal processing.
Antimigration agents may be incorporated at 0.1-0.5% levels to reduce pigment migration during high-temperature processing and use. These agents improve pigment-polymer interaction and reduce the tendency of pigment molecules to diffuse through the polymer matrix.
Processing aids must be selected for high temperature stability, avoiding materials that may decompose or produce volatile compounds at elevated temperatures. Silicone-based processing aids and high-melting-point waxes are preferred for maintaining processability without introducing low molecular weight components that could migrate.
Production Process
The production process for low migration masterbatch demands strict process control, consistent mixing quality, and comprehensive quality verification. Every step must be performed with careful attention to regulatory requirements and product quality.
Raw Material Qualification
The process begins with comprehensive raw material qualification. Pigments must be tested for heavy metal content, residual solvent levels, and compliance with applicable regulations. For food contact applications, raw material safety data sheets (SDS) must confirm approval status under relevant regulatory guidelines.
Carrier polymers must meet purity requirements with low residual monomer content and specified melt flow characteristics. Virgin material is typically required for food contact and medical device applications, though specific regulatory exemptions may allow limited recycled content under certain conditions.
All additives including dispersants, stabilizers, and processing aids must be approved for the intended application category. Certification of compliance from additive suppliers is essential, and traceability documentation should be maintained for all incoming raw materials.
Raw material testing procedures include moisture content analysis, melt flow index measurement, heavy metal testing using atomic absorption spectroscopy, and residual solvent analysis using gas chromatography. Only materials meeting strict acceptance criteria should be used in low migration production.
Formulation Preparation
Formulation preparation involves precise weighing and proportioning of raw materials according to formulation specifications. Automated batching systems are strongly recommended for low migration production to ensure consistent formulation accuracy and prevent cross-contamination.
For sensitive formulations requiring precise pigment loading (±0.1%), gravimetric batching systems provide the necessary accuracy. Batch records should document all raw material lot numbers, weights, and batching parameters for complete traceability throughout production.
Pre-mixing may be performed using low-shear tumble blenders to improve initial distribution of pigment and additives before extrusion. However, high-shear mixing should be avoided to prevent additive degradation or particle size reduction that could increase migration potential.
Material transfer systems must be carefully designed to prevent contamination risks between formulation batches. Closed transfer systems with inert gas purging are recommended for particularly sensitive applications. Material contact surfaces should be made from non-reactive materials and cleaned thoroughly between batches.
Continuous Extrusion Process
The extrusion process for low migration masterbatch must balance effective mixing with minimal thermal degradation and mechanical stress on additives. Continuous twin screw extrusion provides the controlled processing environment necessary for consistent mixing quality.
Feed system accuracy is critical for maintaining formulation consistency throughout continuous production. Gravimetric feeding systems with loss-in-weight technology ensure precise material delivery at consistent rates, preventing concentration variations that could affect migration properties.
Extrusion temperature profile must be optimized for the specific formulation, balancing complete polymer melting with minimal additive thermal degradation. Generally, temperature increases gradually along the barrel length to minimize shear heating and thermal stress. Typical profiles for polyolefin carriers range from 150°C at feed zone to 220°C at die.
Screw configuration should be designed for distributive mixing rather than intensive dispersive mixing to minimize additive particle size reduction that could increase migration tendency. Wider kneading blocks and larger clearance mixing elements provide effective distribution without excessive shear.
Process Monitoring and Control
Continuous process monitoring ensures consistent quality throughout production. Key parameters monitored include barrel temperatures, melt pressure, screw speed, feed rate consistency, and power consumption. Deviations from set points trigger automatic alarms and corrective action procedures.
Advanced control systems provide real-time data analysis and process optimization capabilities. Integration with upstream batching systems and downstream quality control creates a closed-loop quality management system that maintains consistent formulation and processing parameters.
In-line quality measurement technologies including near-infrared (NIR) spectroscopy and color measurement systems enable real-time monitoring of formulation consistency. These systems provide immediate feedback on pigment concentration variations, allowing automatic feed rate adjustments to maintain target formulation.
Process data collection systems store detailed production data for quality traceability and continuous improvement initiatives. Data can be analyzed to identify production trends, optimize process parameters, and implement predictive maintenance strategies.
Quality Control and Testing
Comprehensive quality control protocols ensure compliance with regulatory requirements and product specifications. Testing includes visual inspection, color measurement, migration testing, physical property evaluation, and microbiological analysis where required.
Color measurement using calibrated spectrophotometers ensures color consistency within specified tolerances. Delta E values should typically be maintained below 0.8 for critical applications to meet customer color requirements.
Migration testing represents the most critical quality control step for low migration masterbatch. Methods include overall migration (OM) testing according to ISO 10993-19 and specific migration (SM) testing for individual components. Testing should be performed using food simulants appropriate for intended use conditions.
Physical property testing includes melt flow index measurement, mechanical property evaluation using test plaques, and dispersion quality assessment through microscopic examination. All test results should be documented in batch records for regulatory compliance purposes.
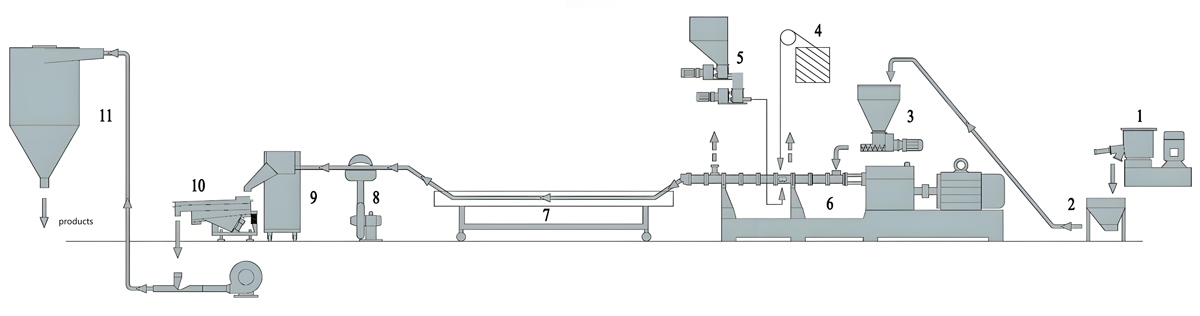
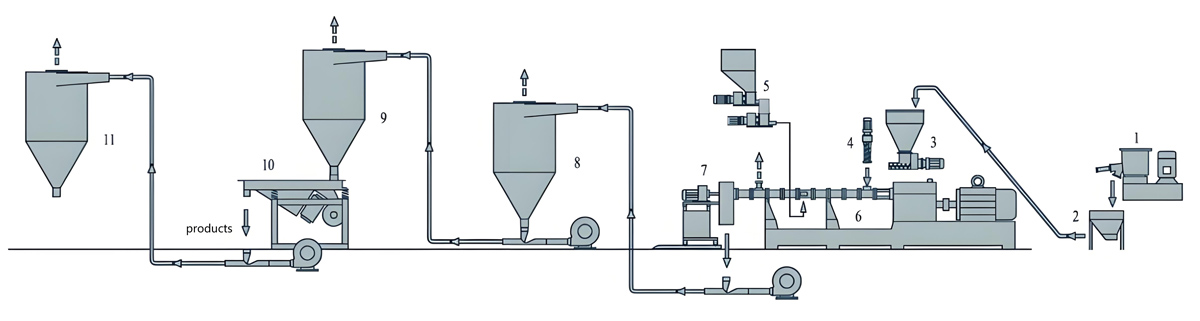
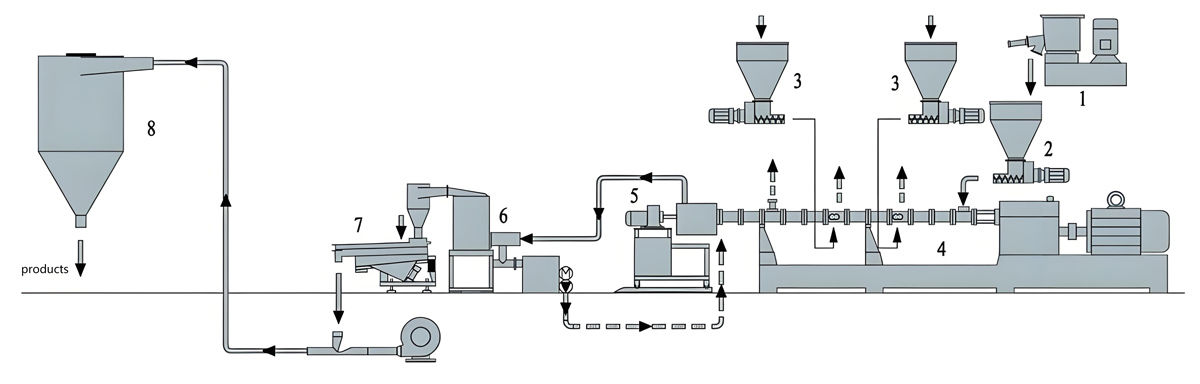
Production Equipment Introduction
Kerke KTE Series Continuous Twin Screw Extruder
The Kerke KTE Series twin screw extruder offers advanced continuous extrusion technology optimized for low migration masterbatch production. Specifically designed for compounding applications requiring precise control, the KTE Series provides consistent mixing quality while minimizing thermal degradation risks. The modular design allows configuration flexibility to meet specific process requirements.
The KTE Series features co-rotating twin screw design with screw diameters from 25mm to 100mm and length-to-diameter ratios from 36:1 to 48:1. This extended processing length provides sufficient residence time for thorough mixing while minimizing shear heating. Barrel segments incorporate precise temperature control zones with individual PID controllers to maintain uniform heat distribution.
Variable speed drive technology enables independent screw speed control from 50-600 rpm depending on machine size. The AC vector drive provides excellent torque characteristics at all speeds, allowing operators to optimize shear levels for specific formulations. Precise speed control is essential for maintaining consistent migration properties across different production runs.
Advanced screw configurations are available for low migration applications, focusing on distributive rather than intensive mixing. The KTE Series includes specialized kneading block geometries and mixing elements designed to achieve uniform distribution without reducing additive particle size to levels that increase migration tendency.
Feeding System
Precise feeding is critical for maintaining formulation consistency and migration resistance in continuous low extrusion production. Multiple feeding configurations can be employed depending on formulation complexity and material characteristics.
Gravimetric feeding systems with loss-in-weight technology are recommended for all low migration applications. These systems provide continuous weight measurement and automatic feed rate adjustment to maintain accurate formulation ratios. Accuracy within ±0.1% is achievable with properly calibrated systems.
For sensitive pigment feeding requiring minimal contamination risk, closed transfer systems with inert gas purging prevent oxidative degradation and cross-contamination risks. Material contact surfaces should be made from polished stainless steel to minimize pigment adhesion and facilitate thorough cleaning.
Feeder calibration procedures must be performed regularly to maintain accuracy. Calibration should be validated using actual production materials rather than standard reference materials to account for specific material flow characteristics. Calibration records should be maintained for regulatory compliance purposes.
For formulations with multiple components requiring precise proportioning, modular feeding systems with individual gravimetric feeders ensure accurate component delivery rates. Centralized control systems coordinate feeder operation to maintain formulation balance throughout production.
Temperature Control System
Precise temperature control is essential for low migration production to ensure complete polymer melting without causing additive thermal degradation. The KTE Series incorporates advanced temperature management systems for consistent heat control.
Zone-based temperature control provides individual PID regulation for each barrel section, allowing precise temperature profile optimization. Barrel heating elements use high-efficiency cartridge heaters with rapid response time, while water cooling jackets provide effective temperature regulation during start-up and shutdown cycles.
Melt temperature monitoring at strategic barrel positions ensures actual melt conditions match set points. Thermal couples placed in melt flow channels provide direct melt temperature measurement rather than relying solely on surface temperature readings.
Control algorithms optimize heating and cooling cycles to minimize temperature fluctuations and ensure uniform melt conditions throughout production. Temperature limits are established for each formulation to prevent overheating that could affect additive stability or migration properties.
Quality Assurance Equipment
Specialized quality assurance equipment is required for low migration production to verify compliance with regulatory standards and product specifications.
Migration testing equipment includes vacuum ovens for overall migration testing according to ISO 10993-19, Soxhlet extraction systems for specific additive analysis, and gas chromatography-mass spectrometry (GC-MS) for quantifying individual migratory compounds.
Color measurement systems including spectrophotometers and color matching software ensure color consistency across production batches. Computerized color analysis provides objective color data that can be correlated with formulation and processing parameters for continuous improvement.
Material characterization equipment includes melt flow index testers, tensile testing machines, and dynamic mechanical analyzers for evaluating physical properties and formulation consistency. These tools help identify formulation changes or process variations that could affect migration resistance.
Parameter Settings
Optimal parameter settings for low migration production depend on formulation specifics, application requirements, and equipment configuration. The following sections provide general guidance, though specific formulations should be validated through experimental optimization.
Extrusion Parameters
Screw speed is typically set to achieve adequate mixing while minimizing shear energy input. For low migration formulations requiring gentle mixing, speeds range from 100-200 rpm depending on screw diameter. Higher speeds may be necessary for formulations with challenging dispersion requirements but should be balanced against potential additive degradation risks.
Temperature profile must be tailored to the specific carrier polymer and additive stability characteristics. For polyolefin-based formulations, feed zone temperature starts at 140-150°C, increasing gradually to 180-190°C in compression zones, 200-210°C in mixing zones, and 210-220°C at the die. Lower temperature profiles may be required for particularly heat-sensitive additives.
Throughput rate depends on machine size and formulation viscosity. For 50mm extruders, typical rates range from 50-150 kg/h for polyolefin formulations. Higher rates may reduce residence time and mixing quality, while lower rates increase thermal exposure time. The balance between throughput and mixing must be optimized for each application.
Back pressure should be maintained at 1.5-2.5 MPa to ensure adequate melt compression and uniform flow through the die. Lower pressures may cause inconsistent pellet quality, while excessively high pressure increases energy consumption and thermal load on additives.
Feeding System Parameters
Feeding rates must be carefully calibrated to maintain accurate formulation ratios throughout production. For multi-component systems, individual feeder rates are synchronized to maintain target proportioning based on formulation specifications.
Feeder response times must be matched to extrusion process dynamics to prevent formulation imbalances during start-up or rate changes. Rapid response gravimetric systems minimize transition material waste while maintaining formulation accuracy.
Material level control in hoppers prevents starvation or overfeeding conditions that could disrupt formulation consistency. Continuous level monitoring using non-contact sensors ensures stable material flow to extruder feed zones.
For hygroscopic materials requiring dry processing, dehumidifying dryers with dew point control (-40°C or lower) ensure consistent moisture content. Moisture levels above 0.1% can cause processing issues and affect migration resistance in finished masterbatch.
Mixing System Parameters
Mixing element configuration depends on formulation mixing requirements. For low migration applications emphasizing distributive mixing, wider kneading blocks (2-3 mm thick) and larger clearance mixing elements are preferred over intensive dispersive mixing elements.
Mixing zone location should be optimized to provide sufficient residence time for uniform distribution without prolonged thermal exposure. Mixing elements are typically placed in the later barrel sections after complete polymer melting.
Shear rate balance is critical for low migration production. Mixing intensity must achieve uniform distribution without causing additive particle size reduction or generating excessive shear heat that could affect additive stability.
Screw design should include barrier elements to separate solid conveying and melting zones from mixing and devolatilization zones. This improves process stability and prevents premature mixing of partially melted polymer with additives.
Equipment Price
The investment required for low migration production depends on production scale, automation level, and quality assurance requirements. The following price estimates are provided for reference purposes and reflect typical ranges for various equipment configurations.
KTE Series Twin Screw Extruder Pricing
The Kerke KTE Series offers competitive pricing while maintaining high quality standards for low migration applications. Price levels vary by machine size and configuration complexity:
KTE-25 (25mm screw diameter): $90,000 – $120,000. Laboratory/pilot-scale model suitable for formulation development and small-scale production.
KTE-40 (40mm screw diameter): $160,000 – $200,000. Mid-range production model offering capacity of 50-150 kg/h with basic process control features.
KTE-50 (50mm screw diameter): $230,000 – $290,000. Standard production model suitable for medium-scale low migration production with advanced control systems.
KTE-75 (75mm screw diameter): $360,000 – $460,000. High-capacity production model for large-scale operations with integrated automation and quality control systems.
Custom configurations with specialized quality assurance equipment and advanced control capabilities may increase prices by 20-30% above standard model prices.
Accessory Equipment Pricing
Essential accessory systems contribute to total investment but are critical for maintaining low migration quality standards:
Gravimetric feeding system with loss-in-weight technology: $30,000 – $70,000 depending on number of feeding stations and complexity requirements.
Migration testing laboratory equipment: $80,000 – $150,000 including vacuum ovens, GC-MS, and related analysis software for comprehensive migration analysis capabilities.
Automated batching system with traceability features: $50,000 – $100,000 for closed-loop formulation management and compliance documentation.
Continuous quality monitoring system with NIR spectroscopy: $40,000 – $80,000 for real-time formulation consistency measurement and process control.
Complete low migration production line with KTE-50 extruder and essential accessories: $380,000 – $500,000 installed, depending on specific configuration complexity and automation level.
Operating Cost Considerations
Beyond initial capital investment, ongoing operating costs significantly impact total cost of ownership. Key operating cost factors include raw material costs, energy consumption, maintenance expenses, and regulatory compliance costs.
Energy consumption varies by machine size and operating conditions. A KTE-50 extruder typically consumes 60-100 kW/h during production, translating to $9,000-$17,000 in annual electricity costs at typical industrial rates assuming 5000 operating hours per year.
Maintenance costs average 2-3% of equipment value annually when following preventive maintenance protocols. This includes routine inspection, wear part replacement, control system calibration, and quality assurance equipment servicing.
Regulatory compliance costs including testing and documentation represent a significant portion of operating costs for low migration production. Ongoing testing for migration and biocompatibility requirements should be budgeted at 0.5-1.0% of annual sales revenue for most applications.
Raw material costs are generally higher for low migration production due to specialized material requirements. Food-grade pigments and additives can cost 200-300% more than standard equivalents, and virgin carrier polymer is typically required for regulatory compliance.
Production Problems and Solutions
Low migration production presents unique challenges requiring specialized troubleshooting approaches. Issues often relate to formulation stability, process control, and regulatory compliance. The following sections address common problems and recommended solutions.
Problem 1: Migration Test Failures
Migration test failures represent the most critical quality issue in low migration production, potentially requiring product recall and regulatory intervention. Failures typically occur when test results exceed specific migration limits established by regulatory agencies.
Cause Analysis
Multiple factors can contribute to migration test failures: incorrect formulation design using unapproved or incompatible materials, excessive additive particle size reduction during processing, inadequate polymer wetting leading to poor additive retention, contamination from cross-material transfer, or thermal degradation of additives during processing creating low molecular weight migration products.
Poor mixing quality can create areas with higher additive concentration that migrate more readily. Thermal degradation during processing can break additive molecules into smaller fragments with increased migration tendency. Incompatible formulation components may phase separate during aging, creating pathways for accelerated additive migration.
Testing methodology can also influence results. Improper sample preparation, inadequate extraction conditions, or analytical method deficiencies can lead to misleading results. Testing should always be performed using validated procedures consistent with regulatory guidelines.
Solution
Address migration failures systematically through root cause analysis. First, verify formulation approval status of all raw materials through supplier documentation and regulatory cross-reference. Replace any unapproved materials with alternatives meeting relevant compliance requirements.
Review extrusion process parameters to identify potential thermal degradation sources. Reduce peak temperatures or increase cooling efficiency to minimize thermal stress on temperature-sensitive additives. Optimize screw configuration to reduce shear energy input while maintaining necessary mixing quality.
Implement improved quality control procedures to identify formulation inconsistencies before full-scale production. Batch-to-batch sampling and testing can catch migration issues early, preventing significant product waste and regulatory non-compliance.
Review test methodology and ensure compliance with official analytical procedures. Verify sample preparation, extraction conditions, and analytical equipment calibration before accepting test failure results as definitive.
Prevention
Implement comprehensive raw material qualification protocols including pre-approval testing for migration potential. Document all material approvals and maintain supplier certification records for regulatory audit readiness.
Establish standard operating procedures emphasizing process conditions that minimize additive degradation and particle size reduction. Train operators on proper process optimization techniques specific to low migration production requirements.
Implement real-time quality monitoring systems to identify formulation consistency issues before they become critical migration problems. Early detection through statistical process control reduces the risk of non-compliant products reaching the market.
Conduct periodic validation studies to confirm ongoing compliance with regulatory requirements as formulations or processes evolve. Validation protocols should include worst-case scenario testing to ensure consistent compliance across product variations.
Problem 2: Inconsistent Migration Properties Between Batches
Inconsistent migration test results between different production batches create quality control challenges and potential supply chain disruptions. This issue occurs when migration test results vary significantly between batches of the same formulated product.
Cause Analysis
Variations in raw material characteristics represent the most common cause of inconsistent migration properties. Differences in pigment particle size distribution, additive purity levels, or carrier polymer molecular weight distribution can affect migration rates even when formulation ratios remain constant.
Process parameter variations also contribute to inconsistent migration properties. Changes in temperature profile, screw speed, or mixing quality can alter additive particle size distribution and dispersion uniformity, affecting migration tendency.
Testing variation from differences in sample preparation, extraction conditions, or analytical measurement can create apparent batch inconsistency that does not reflect actual product differences. Laboratories should implement standardized test procedures and regular calibration checks to minimize variability.
Cross-contamination risks from incomplete material changeover can introduce foreign materials with different migration properties, affecting batch-to-batch consistency. Inadequate cleaning procedures between different formulation batches can cause subtle contamination affecting migration test results.
Solution
Improve raw material consistency through tighter supplier specifications and incoming inspection testing. Request raw material analysis reports from suppliers and perform independent verification testing before production use.
Implement process control systems to maintain consistent extrusion parameters across all production runs. Advanced control algorithms can compensate for minor raw material variations while maintaining consistent process outcomes.
Optimize changeover procedures to prevent cross-contamination risks. Implement dedicated production lines for sensitive product categories when feasible. Document cleaning validation procedures and maintain audit trails for material changeover activities.
Standardize test procedures across all testing locations and maintain ongoing proficiency testing programs to minimize measurement variability. Calibrate analytical equipment regularly and participate in external quality assessment programs to verify analytical accuracy.
Prevention
Implement statistical process control (SPC) programs to monitor migration test results over time and identify trends indicating developing consistency issues. Control charts help distinguish between random variation and systemic process problems requiring intervention.
Develop robust changeover protocols including residue testing to confirm complete removal of previous materials before new batch production. Use validated rinse procedures and limit cross-use of common equipment between sensitive product groups.
Maintain comprehensive raw material qualification records and perform periodic re-qualification assessments of critical components. Update supplier specifications based on ongoing material performance evaluation in production.
Conduct inter-laboratory comparison testing to verify consistent test results across different testing locations. This establishes baseline performance data and identifies potential laboratory-specific bias issues affecting migration test results.
Problem 3: Poor Dispersion Affecting Color Uniformity
Poor pigment dispersion causes color inconsistency and potentially higher migration rates due to locally high pigment concentrations. Dispersion issues often manifest as color streaks, specks, or uneven color distribution in finished masterbatch.
Cause Analysis
Inadequate wetting of pigment particles by polymer melt can result from insufficient temperature, low screw speed, or improper dispersant selection. Incompatible dispersants fail to reduce pigment agglomerate forces effectively.
Excessive shear can cause pigment particle fragmentation, increasing surface area and potentially enhancing migration tendency. However, insufficient shear energy leaves pigment agglomerates intact, creating uneven distribution that affects both color and migration properties.
Mixing configuration may lack sufficient distributive mixing elements to achieve uniform pigment distribution. Screw design should balance dispersion quality with minimal particle size reduction for low migration applications.
Inconsistent feeding can cause local pigment concentration variations. Inaccurate feeders or material bridging disrupts formulation balance during continuous production runs.
Solution
Optimize extrusion temperature profile to ensure sufficient polymer viscosity reduction for effective pigment wetting while maintaining additive stability. Increase melt temperature 10-20°C if poor wetting is confirmed through microscopic examination.
Adjust screw speed to provide sufficient shear energy for dispersion while minimizing particle fragmentation. For low migration applications requiring gentle mixing, increase speed incrementally to find balance between dispersion quality and particle size preservation.
Revise screw configuration to add distributive mixing elements. Wider kneading blocks (3-4mm) provide effective distribution while minimizing shear intensity that could reduce particle size and increase migration potential.
Optimize dispersant type and concentration for specific pigment characteristics. Increase dispersant level up to 2% if wetting improvement is needed, ensuring new levels remain within regulatory compliance limits.
Prevention
Implement incoming pigment quality control testing including particle size distribution and surface area analysis. Reject material outside specified limits to prevent dispersion issues before production begins.
Establish dispersion quality baseline data for critical formulations using standardized microscopic examination methods. Monitor trends to identify gradual changes in pigment dispersion characteristics requiring process adjustments.
Document and follow standard operating procedures for screw configuration based on formulation type. Use configuration templates to ensure consistent mixing approach across different batches of similar formulations.
Calibrate feeding systems regularly and implement material flow monitoring to detect bridging or uneven feed patterns that could disrupt formulation consistency. Automated alarm systems alert operators to potential feed issues before they affect dispersion quality.
Problem 4: Extrusion Instability Affecting Process Consistency
Extrusion instability causes process variations that can affect migration properties and product consistency. Instability may manifest as pressure fluctuations, throughput variations, or melt quality irregularities affecting finished product quality.
Cause Analysis
Material feed irregularities from poorly calibrated feeders or bridging problems cause inconsistent material delivery rates. Variable feed rates disrupt extrusion equilibrium conditions, causing fluctuations in pressure and melt quality.
Temperature control issues including inconsistent heater performance or inadequate cooling capacity cause melt temperature variations. This disrupts material viscosity profile and can affect polymer melting uniformity and additive dispersion.
Screw configuration deficiencies such as poor melting zone design or inadequate compression ratio can cause incomplete polymer melting, creating two-phase flow conditions leading to instability. Inconsistent melting requires constant adjustment of operating parameters to maintain process equilibrium.
Inadequate degassing system performance allows trapped air or moisture to remain in melt, causing foam formation and irregular flow patterns through die. Poor venting also disrupts formulation stability by changing melt volume characteristics unpredictably.
Solution
Improve feed system performance through regular calibration and maintenance. Implement level control in material hoppers to prevent starvation and use vibratory agitation to minimize bridging tendency of poorly flowing materials.
Upgrade temperature control system using high-precision heaters and efficient cooling circuits. Implement redundant temperature sensors and fail-safe control algorithms to maintain stable temperature profiles under varying process conditions.
Optimize screw configuration for melting performance using barrier flight technology to separate melting and solid conveying zones. This improves melting efficiency and provides more consistent melt quality across operating ranges.
Enhance degassing system performance with secondary vents or improved vacuum pump capacity. Maintain vacuum level above 700 mmHg for effective removal of volatile compounds and moisture from melt.
Prevention
Implement preventive maintenance program with scheduled equipment inspection and calibration intervals. Regularly monitor wear indicators on screw elements and barrel surfaces to maintain consistent clearance dimensions critical for process stability.
Implement process control algorithms designed to compensate for process disturbances automatically. Advanced control systems using model predictive control (MPC) can anticipate and counteract potential instability sources before they affect production quality.
Maintain detailed process parameter logs to identify trends indicating developing instability issues. Statistical analysis of production data can detect subtle changes before they become critical process disruptions.
Train operators to recognize early instability signs through real-time parameter monitoring and respond through established standard operating procedures. Operators should understand root cause analysis techniques for common instability sources.
Problem 5: Thermal Degradation of Additives
Thermal degradation during processing can break additive molecules into smaller fragments with increased migration potential and reduced effectiveness. Degradation products may also create safety concerns for food contact applications.
Cause Analysis
Excessive processing temperature, prolonged residence time at high temperature, or shear-induced heating can exceed additive thermal stability limits, causing molecular breakdown. Inadequate cooling capacity may allow melt temperature to exceed set points under load conditions.
Oxidative degradation occurs when heat-sensitive additives react with oxygen in the processing environment. Poor material transfer system design may introduce air into melt stream, increasing oxidation potential at elevated temperatures.
Contamination sources including residual cleaning agents or previous formulation components can catalyze additive degradation reactions. Inadequate purge procedures between formulation changeovers introduce reactive species that affect additive stability.
Thermal cycling during start-up/shutdown sequences can subject additives to repeated temperature changes accelerating degradation over multiple production runs. Poor process control during transition periods can cause excessive thermal stress on sensitive materials.
Solution
Reduce peak temperature profile by 10-20°C while maintaining adequate polymer melting. Optimize temperature gradients to minimize shear heating effects particularly in mixing and compression zones of extruder.
Implement oxygen-free processing environment through inert gas purging of material transfer systems and feed zones. Nitrogen blanketing reduces oxidative degradation risks for particularly heat-sensitive additives.
Improve transition period control through automated start-up and shutdown procedures that minimize thermal cycling effects. Implement controlled temperature ramp sequences rather than rapid temperature changes during transitions.
Upgrade heater control systems for more precise temperature regulation. Use proportional-integral-derivative (PID) control algorithms with optimized tuning parameters for each zone based on formulation thermal sensitivity.
Prevention
Validate additive stability under actual processing conditions through accelerated testing protocols. Document temperature limits and maximum residence times for each formulation component to establish operational boundaries.
Implement automated process safety interlocks that reduce temperature set points during process upsets or when production rates drop below minimum thresholds where residence time increases substantially.
Establish changeover procedures emphasizing thorough equipment purging using compatible materials. Record purge times and material quantities for each changeover to ensure consistent cleaning effectiveness.
Monitor melt quality characteristics online using advanced sensors to detect degradation products early. Near-infrared spectroscopy can identify chemical changes indicating additive breakdown before critical limits are exceeded.
Maintenance
Proper maintenance is critical for maintaining consistent low migration quality standards and equipment longevity in continuous production environments. Preventive maintenance programs should address wear component replacement, control system calibration, safety equipment testing, and process validation protocols.
Preventive Maintenance Schedule
The following maintenance schedule provides recommended intervals for low migration production equipment. Adjustments may be needed based on specific operating conditions and formulation requirements.
Daily Maintenance
Operators should perform routine daily checks including visual inspection of machine for leaks, unusual sounds, or vibration. Temperature and pressure gauge readings should be verified against set points and logged in batch records for compliance documentation.
Feed system inspection includes verifying material flow condition, checking hopper levels, and confirming proper feeder calibration status. Clean feed zones if excessive material build-up is observed that could affect consistent feeding.
Calibrate online quality sensors using reference standards at beginning of production shifts. Color measurement systems should be checked against certified color standards daily to ensure measurement accuracy.
Record energy consumption data for each production run to track operational efficiency trends over time. Significant increases in energy use may indicate developing equipment wear that requires attention.
Weekly Maintenance
Weekly maintenance includes detailed inspection of wear components including screw elements and barrel surfaces. Measure wear indicators on critical mixing sections using precision measuring tools.
Calibrate gravimetric feeding systems using standardized weight verification procedures. Check feeder accuracy across operational range to ensure consistent formulation ratio maintenance.
Inspect temperature control systems including sensor calibration status and heater performance. Clean heating element insulation covers to maintain heat transfer efficiency and prevent overheating hazards.
Inspect vacuum degassing system performance including pump oil level and filter condition. Replace filters and perform pump maintenance according to manufacturer recommended intervals.
Monthly Maintenance
Monthly maintenance includes comprehensive inspection of drive system components including motor bearings, gearbox, and coupling condition. Check gear lubrication level and quality, replenish or change oil as needed.
Validate process control system performance through simulated process deviation tests. Verify alarm response times and safety interlock functionality according to equipment safety documentation.
Inspect quality assurance laboratory equipment including GC-MS calibration status and migration testing oven performance. Review analytical method validation documentation for ongoing compliance with regulatory requirements.
Clean material transfer system components and perform leak testing of closed transfer lines. Verify inert gas supply purity level meets specified standards for low oxygen processing environments.
Quarterly Maintenance
Quarterly maintenance involves major wear component inspection and potential replacement. Remove screw elements and check wear patterns and dimensional accuracy using precision measurement equipment.
Perform complete electrical system safety audit including ground continuity testing, motor winding resistance measurement, and control panel enclosure integrity check according to safety standards.
Validate migration testing procedures through inter-laboratory comparison testing or participation in proficiency testing programs. Update analytical method documents based on test performance review.
Review maintenance records and update preventive maintenance schedule based on observed equipment wear rates and formulation-specific operational demands. Adjust frequency based on actual performance data.
Annual Maintenance
Annual preventive maintenance includes complete machine disassembly and component inspection at major service intervals. Evaluate wear condition of screw elements, barrel liners, bearings, seals, and drive system components.
Validate calibration of all control system instruments using traceable reference standards. Document calibration records for regulatory audit purposes including instrument serial numbers, calibration dates, and adjustment history.
Perform comprehensive safety system validation including emergency stop functionality, guard interlocks, and pressure relief valve testing. Review safety documentation update status based on regulatory requirements changes.
Review complete maintenance program effectiveness and adjust based on equipment performance data. Implement continuous improvement initiatives based on downtime analysis and maintenance cost trends.
FAQ
Q1: What materials are considered low migration for food contact applications?
Low migration materials for food contact applications must comply with FDA indirect food additives regulations (21 CFR) and EU 10/2011 plastic materials regulation. Approved materials include FDA-listed pigments meeting 21 CFR 178.3297, food-grade carrier polymers meeting 177.1520 for olefins, and additives authorized under appropriate sections of the food additive regulations.
Key material selection criteria include molecular weight above 1000 Da, good polymer compatibility, low volatility at processing temperatures, and demonstrated resistance to migration under intended use conditions. High molecular weight additives have reduced diffusion rate through polymer matrix compared to low molecular weight alternatives.
Material suppliers should provide certificates of compliance confirming approval status. Third-party testing agencies can verify migration compliance through specific migration testing according to regulatory protocols. Always consult with regulatory experts for complex formulation approval status determination.
Common approved pigment types include titanium dioxide (R104, R-960), phthalocyanine blues and greens, quinacridone reds and violets, and selected azo pigments approved under 21 CFR 178.3297. However, specific approval status depends on exact chemical composition and impurity levels.
Q2: How does processing temperature affect migration potential?
Temperature significantly affects migration properties through two primary mechanisms: temperature increases mobility of polymer chains, creating pathways for additive molecules to diffuse through the matrix; and high temperatures can cause additive degradation, forming smaller fragments with increased migration tendency.
Maintaining processing temperatures at minimum required for polymer melting reduces polymer chain mobility and minimizes thermal degradation risks. Typical temperatures range 180-220°C for polyolefin carriers, though specific values depend on polymer melting characteristics and additive stability.
Localized hot spots in the extruder may exceed average set points, causing increased degradation at those locations. Advanced temperature control systems monitor multiple points along barrel length to prevent this potential issue.
Post-processing temperature history also affects migration during service life. Sterilization or pasteurization processes for food containers can increase additive diffusion rate temporarily until temperature returns to ambient conditions.
Q3: What testing methods are used to verify migration compliance?
Regulatory migration testing follows standardized protocols including overall migration (OM) testing according to ISO 10993-19 and specific migration (SM) testing for individual substances according to relevant national or international standards.
Overall migration determines the total amount of material that migrates from polymer to food simulant under specified conditions. Test involves immersing specimen in appropriate food simulant at defined temperature for set time, then measuring non-volatile residue left after solvent evaporation.
Specific migration testing quantifies migration levels of individual substances such as pigments, stabilizers, or plasticizers. These tests require analytical methods capable of detecting and quantifying target analytes at parts per million (ppm) or parts per billion (ppb) levels depending on substance-specific migration limits.
Testing conditions are selected based on intended use conditions including contact time, temperature, and food type represented by appropriate food simulant (water, 3% acetic acid, 50% ethanol, or isooctane depending on regulatory jurisdiction).
Q4: How do I choose the right screw configuration for low migration masterbatch?
Screw configuration should prioritize distributive rather than intensive dispersive mixing to minimize additive particle size reduction that could increase migration potential. Focus on achieving uniform additive distribution without breaking down pigment agglomerates to molecular levels where migration tendency increases.
Recommended configuration starts with conveying elements in feed section, transition to melting zone incorporating barrier flight technology to separate solid and melt phases, then distribution zone with wide kneading blocks (3-4mm thick) and neutral mixing elements to achieve uniform additive distribution.
Reverse conveying elements can be used sparingly to increase residence time without excessive shear. Screw length-to-diameter ratios of 36:1 to 40:1 generally provide sufficient processing length for thorough mixing while maintaining reasonable residence time.
Clearance between screw elements and barrel should be sufficient to minimize shear stress on additives while maintaining mixing efficiency. Typical clearances range from 0.5-1.0mm depending on screw diameter and formulation viscosity characteristics.
Q5: What are the typical changeover procedures for low migration production?
Changeover procedures for low migration applications emphasize thorough cleaning to prevent cross-contamination risks between formulations. The process begins with emptying all material handling systems including feed hoppers, pipes, and extruder barrel.
Purge material is introduced to clean extruder barrel and screw elements. For most formulations, a compatible grade of the new carrier polymer can serve as purge material. However, specialized color change purge compounds may be needed for difficult transitions.
Cleaning verification involves checking extrudate visual appearance for traces of previous color/formulation and collecting samples for analytical testing if required by regulatory protocols. Transition material is typically segregated and reprocessed if formulation compatibility allows.
For particularly sensitive applications, equipment disassembly and mechanical cleaning may be required periodically despite normal purge procedures. This involves removing screw elements and barrel liners for manual cleaning using approved procedures to eliminate cross-contamination risks completely.
Q6: How do I ensure ongoing regulatory compliance for low migration masterbatch?
Ongoing compliance requires continuous vigilance across all production stages. Start with maintaining comprehensive regulatory documentation including raw material certificates of compliance, batch records, test reports, and procedure manuals.
Participate in regular regulatory audits both internal and third-party to identify potential compliance gaps before they become critical issues. Keep up to date with regulatory changes through active participation in industry associations and regulatory working groups.
Implement change control procedures that require evaluation of regulatory impact before any process or formulation modification is approved. Document all changes with associated validation studies demonstrating compliance continuity.
Train all production personnel on regulatory requirements specific to low migration applications. Conduct regular training refreshers to ensure ongoing awareness of compliance protocols and documentation requirements.
Q7: What role does polymer molecular weight play in migration resistance?
Polymer molecular weight significantly affects additive migration rate. Higher molecular weight polymers have lower free volume between polymer chains, creating less accessible pathways for additive molecule diffusion through the polymer matrix.
Carrier polymers for low migration applications should have molecular weight distribution optimized for intended use. Narrow molecular weight distributions provide more uniform migration performance compared to broader distributions where lower molecular weight fractions may allow faster additive diffusion.
Crosslinking agents can increase migration resistance by further reducing polymer chain mobility through three-dimensional network formation. However, crosslinking may affect processing characteristics and must be carefully controlled to maintain product performance.
Crystalline polymer regions act as physical barriers to additive diffusion. Higher crystallinity generally improves migration resistance compared to amorphous regions, though total crystallinity depends on polymer type and processing conditions during manufacturing.
Q8: How can I troubleshoot inconsistent migration test results?
Inconsistent results require systematic root cause analysis starting with verification of test methodology accuracy. Confirm testing parameters including temperature, time, food simulant type, specimen preparation, and analytical measurement procedures match regulatory requirements.
Review raw material batch records for potential changes in pigment or additive characteristics between production batches. Pigment particle size distribution changes can affect migration behavior even when formulation loading remains constant.
Analyze process data for variations during production that might affect migration properties such as temperature excursions, screw speed deviations, or feed rate inconsistencies. Statistical correlation analysis can identify potential relationships between process variations and migration test results.
Consider material aging effects as migration properties may change during storage particularly in warm or high-humidity environments. Freshly produced material should be tested alongside aged material to evaluate storage impact on migration characteristics.
Q9: What is the relationship between dispersibility and migration tendency?
Dispersion quality has complex relationship with migration properties. Ideally, pigment should be uniformly distributed as discrete particles throughout polymer matrix. Well-dispersed pigment avoids local concentration peaks that exhibit higher migration rate than average bulk concentration.
Excessive dispersion reducing pigment particle size below certain threshold can increase migration potential by increasing effective surface area exposed to surrounding polymer matrix. Particles smaller than 0.1 μm exhibit different diffusion behavior compared to larger particles, potentially migrating more readily.
Agglomerated pigment may exhibit reduced migration from the large particle because only surface molecules are exposed, but if agglomerates break down during service life (e.g., through mechanical stress), sudden increase in migration may occur.
Dispersant selection affects both dispersion quality and migration behavior. Compatibility between dispersant and polymer matrix is critical for long-term stability. Poorly compatible dispersants may themselves migrate more readily, carrying pigment particles with them or creating pathways for increased pigment diffusion.
Q10: What factors should I consider when selecting suppliers for low migration materials?
Supplier evaluation criteria include: demonstrated compliance with applicable regulatory standards through documented approval status; consistent material quality through ISO 9001 certification and quality management systems; technical support capabilities for formulation development and troubleshooting; and traceability systems to ensure material origin and batch history can be verified.
Suppliers should provide comprehensive technical documentation including safety data sheets (SDS), regulatory compliance certificates, technical specifications, and typical application data. Availability of stability testing data under simulated processing conditions is particularly valuable for low migration application material selection.
Financial stability and supply chain reliability ensure consistent material availability critical for continuous production operations. On-site audit capabilities allow verification of supplier quality systems and manufacturing practices affecting end-product consistency.
Material batch-to-batch consistency should be evaluated through statistical analysis of key properties like particle size distribution, purity levels, and migration test results over multiple lots. Suppliers should provide confidence intervals on critical material characteristics to assist formulation stability prediction.
Summary
Low migration masterbatch manufacturing requires specialized knowledge, dedicated equipment, and strict adherence to quality and regulatory standards throughout production. The continuous twin screw extruder provides the controlled processing environment necessary for consistent mixing quality while minimizing additive degradation and migration risks.
Formulation design represents the foundation of successful low migration production, requiring careful selection of approved materials with good polymer compatibility and minimal migration potential. Material certification and testing ensure compliance with applicable regulatory requirements before they are introduced into the production process.
Processing parameters including temperature, screw speed, and mixing configuration must balance effective dispersion with minimal additive particle size reduction that could increase migration tendency. The KTE Series twin screw extruder provides precise control over these parameters through advanced drive technology and modular configuration capabilities.
Comprehensive quality assurance programs including migration testing, formulation verification, and statistical process control ensure consistent compliance across production runs. Regulatory requirements must be integrated throughout production rather than considered isolated post-production testing steps.
Preventive maintenance programs are essential for maintaining equipment reliability and performance consistency over extended production runs. Regular monitoring of wear components, control system calibration, and safety equipment validation ensures compliance with safety standards and product quality requirements.
By understanding the scientific principles governing additive migration and implementing robust control measures throughout the production process, manufacturers can produce high-quality low migration masterbatch that meets stringent regulatory standards. This requires ongoing commitment to continuous improvement through process optimization, regulatory awareness, and technology upgrades to meet evolving industry demands.
Ultimately, successful low migration production balances regulatory compliance requirements with economic viability through efficient process design, optimized material selection, and effective risk management strategies. By combining advanced extrusion technology with rigorous quality management systems, manufacturers can establish themselves as reliable suppliers of low migration masterbatch in demanding application sectors.