Introduction
Medical grade masterbatch production represents one of the most demanding sectors in plastic compounding due to stringent quality requirements, strict regulatory compliance, and zero tolerance for contamination. Counter rotating twin screw extruders have emerged as preferred equipment for medical grade masterbatch manufacturing due to their superior mixing capabilities, excellent thermal control, and ability to maintain material integrity. The counter rotating configuration provides positive displacement characteristics ideal for handling sensitive medical materials while achieving uniform dispersion of additives and colorants at extremely high quality levels. Nanjing Kerke Extruder Equipment Company KTE Series counter rotating twin screw extruders offer specialized features specifically designed for medical grade applications including sanitary design, corrosion-resistant materials, and precise process control systems.
Medical grade masterbatches serve critical functions in pharmaceutical packaging, medical device components, diagnostic consumables, and other healthcare applications where material purity and consistency are paramount. These masterbatches typically incorporate specialized additives including colorants, UV stabilizers, antimicrobial agents, and other functional additives that must meet biocompatibility requirements and regulatory standards including FDA, USP, and ISO 10993. Counter rotating twin screw extruders provide the gentle yet effective mixing required to maintain the integrity of these sensitive additives while achieving homogeneous distribution throughout the carrier polymer.
The selection of counter rotating twin screw extruder configuration for medical grade masterbatch production requires careful consideration of multiple factors including material compatibility, cleaning requirements, contamination control, validation capabilities, and process documentation. Medical applications demand equipment that can be thoroughly cleaned and validated, with traceability of processing parameters and production conditions. KTE Series counter rotating twin screw extruders from Nanjing Kerke Extruder Equipment Company incorporate design features specifically addressing these medical industry requirements while providing the performance and reliability needed for continuous high-quality production.
Formulation Ratios for Medical Grade Masterbatch
Formulation development for medical grade masterbatch requires balancing multiple performance requirements while maintaining biocompatibility and regulatory compliance. Different medical applications demand specific additive types and concentrations optimized for end-use requirements.
Medical color masterbatch formulations typically incorporate carrier polymer at 80 to 95 percent, colorant pigments at 5 to 20 percent, and processing aids at 1 to 3 percent. Carrier polymer selection typically matches the end-use material including medical-grade polypropylene, polyethylene, polycarbonate, or other medical-grade polymers. Colorant selection for medical applications requires pigments with proven biocompatibility and stability under sterilization conditions. Typical medical colorants include titanium dioxide for white, iron oxides for various earth tones, and carefully selected organic pigments for bright colors. Processing aids including dispersants, flow enhancers, and antistatic agents are selected from FDA-approved materials with appropriate regulatory documentation.
UV stabilizer masterbatch formulations for medical applications generally include UV absorbers at 10 to 30 percent, hindered amine light stabilizers at 5 to 15 percent, processing aids at 1 to 5 percent, with carrier polymer making up the balance to 100 percent. Medical UV masterbatches must maintain effectiveness through sterilization processes and provide long-term protection in medical packaging applications. The UV absorber concentration depends on required protection level and end-use exposure conditions. Higher-end medical packaging for light-sensitive pharmaceuticals may require UV absorber concentrations up to 30 percent while general medical packaging typically uses 10 to 15 percent concentrations.
Antimicrobial masterbatch formulations for medical applications incorporate antimicrobial agents at 1 to 20 percent, depending on required efficacy and end-use requirements. Silver-based antimicrobials typically function at 0.5 to 2 percent active silver content. Zinc-based agents require higher loading levels of 2 to 8 percent. Organic antimicrobials including triclosan alternatives function at 0.5 to 5 percent loading levels. The formulation must balance antimicrobial effectiveness with material properties, processing requirements, and regulatory approval status. Medical device applications requiring long-term antimicrobial activity may use higher loading levels while packaging applications typically use lower concentrations.
Radiopaque masterbatch formulations for medical imaging applications incorporate radiopaque agents at 10 to 40 percent loading levels. Barium sulfate is the most common radiopaque agent for medical applications with typical loading levels of 15 to 35 percent. Bismuth compounds provide alternative radiopaque agents with 10 to 30 percent loading requirements. Higher loading levels provide better radiopacity but may affect material properties and processability. The formulation must balance imaging requirements with material performance, especially for medical device applications where mechanical properties are critical.
Carrier polymer selection for medical grade masterbatch must match end-use material and meet medical grade specifications. Polypropylene medical grades typically require melt flow rates matching end-use material with range from 2 to 50 grams per 10 minutes depending on application. Polyethylene medical grades are selected based on density and melt flow requirements with typical MFR range from 0.5 to 30 grams per 10 minutes. Polycarbonate medical grades require careful selection based on molecular weight and viscosity characteristics matching end-use requirements. Medical grade polystyrene and ABS are selected for specific applications with appropriate flow characteristics.
Production Process for Medical Grade Masterbatch
Medical grade masterbatch production requires controlled processing conditions designed to maintain material purity, achieve uniform additive distribution, and meet strict quality requirements. Counter rotating twin screw extruders provide the gentle yet effective mixing needed for sensitive medical materials.
Material preparation begins with comprehensive material qualification and testing. All incoming materials including carrier polymers, colorants, additives, and processing aids must be accompanied by certificates of analysis confirming identity, purity, and compliance with medical grade specifications. Materials should be tested for moisture content, with most medical polymers requiring drying to less than 0.02 percent moisture content before processing. Medical grade colorants and additives typically require pre-screening for particle size distribution to ensure proper dispersion during processing. Material testing should include analysis for potential contaminants including heavy metals, microbial contamination, and other impurities that could affect medical application safety.
Feeding systems for medical masterbatch production must provide accurate and consistent material delivery. Gravimetric feeding systems with accuracy better than 0.5 percent of setpoint are recommended for medical applications to ensure consistent formulation ratios. Multiple feeders enable separate feeding of carrier polymer, colorants, additives, and processing aids allowing precise control of each component. Loss-in-weight feeders provide continuous weight-based feeding compensating for material density variations. Feeding systems should be designed for easy cleaning and validation with sanitary design features including smooth surfaces, minimal crevices, and clean-in-place capabilities where appropriate.
Extrusion processing for medical grade masterbatch requires precise temperature control typically ranging from 180 to 260 degrees Celsius depending on carrier polymer. Polypropylene medical grades typically process at 190 to 230 degrees Celsius. Medical grade polyethylene processes at 160 to 220 degrees Celsius. Polycarbonate medical grades require higher temperatures of 260 to 300 degrees Celsius. Temperature profiles should be set with gradual heating through feed zone, melting zone, mixing zone, and die zone to ensure uniform melting without thermal degradation. Counter rotating twin screw extruders typically require 8 to 12 heating zones for precise thermal management across the barrel length.
Screw speed optimization for medical masterbatch production balances mixing effectiveness with gentle material handling. Typical screw speeds for medical applications range from 50 to 200 rpm depending on material sensitivity and additive loading. Higher speeds provide better mixing but increase shear heating which may degrade sensitive additives. Lower speeds provide gentler processing but may require longer residence times to achieve adequate mixing. Optimal speed depends on additive types, carrier polymer characteristics, and required dispersion quality. Medical color masterbatches typically use moderate speeds of 80 to 150 rpm providing good mixing without excessive shear.
Vent and devolatilization remove moisture and volatiles that could cause defects in final masterbatch. Medical grade polymers often contain residual moisture despite drying, requiring effective venting during extrusion. Counter rotating twin screw extruders typically include one or more vent ports positioned along barrel length. First vent typically located after melting zone removes moisture and low-boiling volatiles. Second vent positioned near die removes any volatiles from additives or degradation products. Vacuum venting may be used for applications requiring thorough volatile removal. Vent filters prevent particle loss while allowing volatile escape.
Pelletizing system selection affects pellet shape, size, and quality characteristics. Strand pelletizing is commonly used for medical masterbatches providing uniform cylindrical pellets. Strand die design should produce consistent strand diameter with good surface quality. Water bath temperature control prevents thermal shock to hot strands. Pelletizer knives must be sharp and properly aligned for clean pellet cutting. Underwater pelletizing may be used for applications requiring very high pellet quality or handling of shear-sensitive materials. Pellet size typically ranges from 2 to 4 mm in diameter with 2 to 4 mm length for medical applications.
Post-extrusion cooling and handling maintain pellet quality and prevent contamination. Pellets should be cooled to room temperature before packaging to prevent moisture absorption and clumping. Cooling systems should provide uniform cooling without causing pellet deformation or surface defects. Handling systems including conveyors and storage hoppers should be designed for minimal product contact and easy cleaning. Medical grade masterbatch typically requires packaging in sealed containers to prevent contamination and moisture absorption. Packaging materials should be compatible with medical requirements and provide adequate protection during storage and transport.
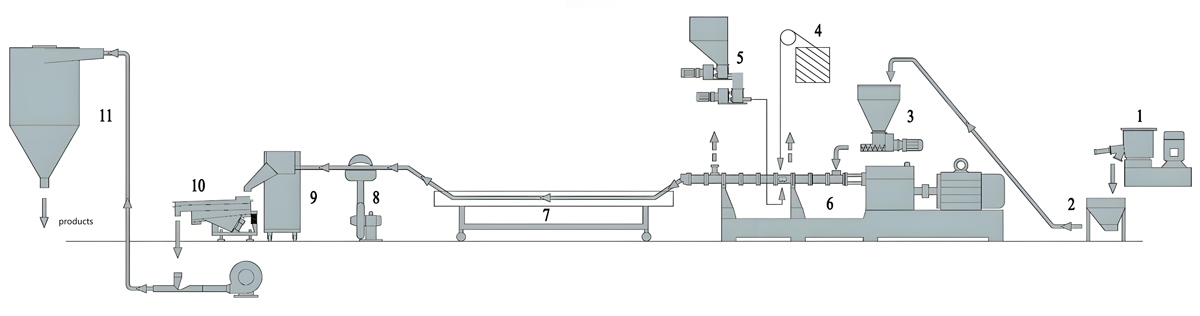
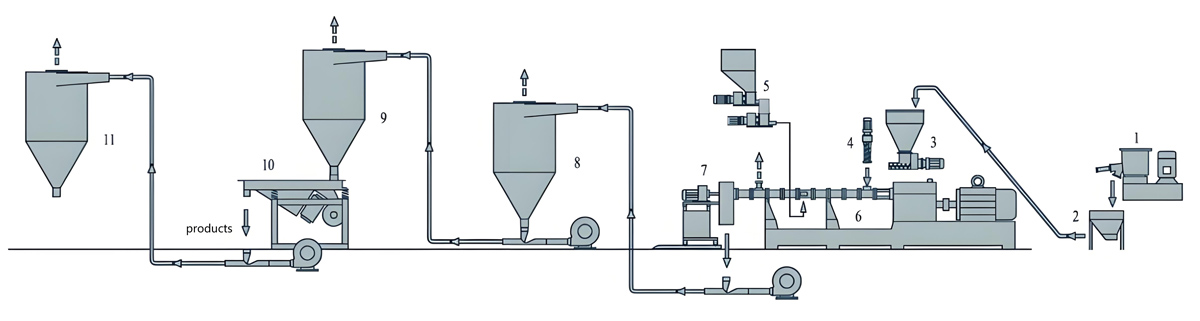
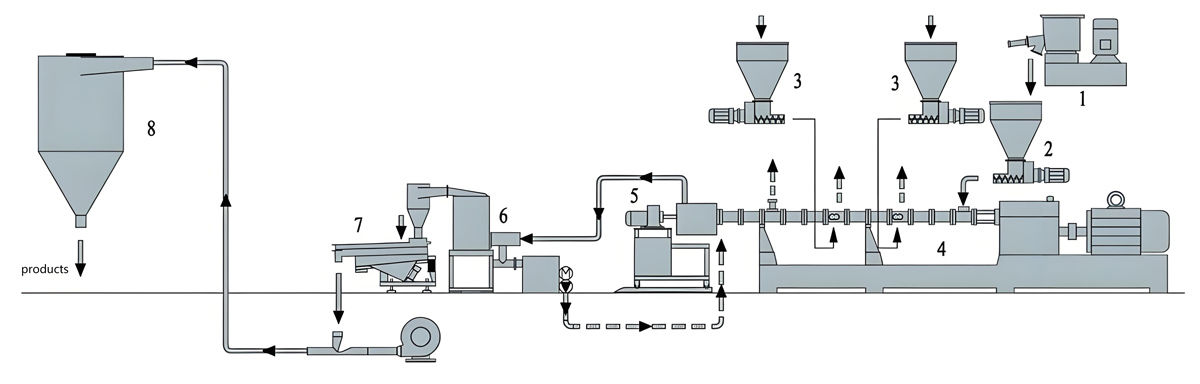
Production Equipment Introduction
Counter rotating twin screw extruders for medical grade masterbatch production incorporate specialized features designed to meet medical industry requirements including contamination control, easy cleaning, and precise process control.
KTE Series counter rotating twin screw extruders from Nanjing Kerke Extruder Equipment Company provide advanced capabilities specifically optimized for medical grade applications. The counter rotating configuration features intermeshing screws rotating in opposite directions providing positive displacement characteristics ideal for sensitive medical materials. This configuration provides excellent mixing while maintaining relatively low shear rates compared to co-rotating designs, making it suitable for shear-sensitive additives and colorants. The positive displacement action ensures consistent material transport through the barrel with minimal material residence time variation.
Barrel and screw construction for medical applications use corrosion-resistant materials meeting medical industry requirements. Standard construction typically uses nitrided or hard-faced steel with additional surface treatments for enhanced corrosion resistance. For highly sensitive applications, barrels and screws can be constructed from stainless steel alloys providing maximum corrosion resistance and cleanability. Screw design features modular construction allowing customized configuration for specific medical masterbatch formulations. Typical medical grade screw designs include specialized mixing elements optimized for gentle dispersion of sensitive additives.
Temperature control systems provide precise thermal management essential for medical material processing. KTE Series extruders feature multi-zone electric heating with individual zone control typically ranging from 8 to 12 zones along barrel length. Heating elements provide capacity up to 10 kW per meter of barrel length ensuring adequate heating even for high-temperature medical polymers. Cooling systems incorporate both air and water cooling providing responsive temperature control during processing. Temperature control accuracy better than plus or minus1 degree Celsius ensures consistent thermal history for medical grade materials. Advanced PID controllers with auto-tuning capabilities maintain optimal temperature profiles.
Drive systems for medical applications provide reliable power with precise speed control. AC vector drives with high-efficiency motors provide 15 to 30 percent energy savings compared to older drive technologies. Drive power ranges from 15 to 400 kW depending on extruder size and processing requirements. Speed control provides adjustment from 10 to 100 percent of maximum speed with accuracy better than 0.1 percent of setpoint. Torque monitoring provides feedback on material conditions and processing consistency. Drive systems include safety features including overload protection and emergency stop functionality.
Control systems for medical masterbatch production provide advanced monitoring and control capabilities. KTE Series extruders feature PLC-based control systems with touch screen interfaces for operator convenience and data access. Process monitoring includes temperature profiles, screw speed, motor load, feed rates, and pressure readings. Data logging capabilities record processing parameters for batch traceability and validation requirements. Alarm systems alert operators to parameter deviations that could affect product quality. Some models offer remote monitoring capabilities for production oversight and technical support.
Sanitary design features facilitate cleaning and validation for medical applications. Equipment surfaces are designed with minimal crevices and smooth finishes to prevent contamination accumulation. Easy-access panels and removable components facilitate thorough cleaning procedures. Some models offer clean-in-place capabilities for automated cleaning cycles. Sanitary design includes rounded corners, polished surfaces, and sloped surfaces promoting drainage. Documentation support includes cleaning protocols, validation guidance, and material compatibility information.
Parameter Settings
Optimal parameter settings depend on specific formulation, carrier polymer, and production requirements. Following parameter ranges provide starting points for medical grade masterbatch production but should be optimized for each specific application.
Temperature profile settings vary based on carrier polymer and formulation requirements. For polypropylene medical masterbatch with 10 percent colorant loading, typical settings include feed zone at 180 to 200 degrees Celsius, compression zone at 200 to 220 degrees Celsius, mixing zone at 210 to 230 degrees Celsius, and die zone at 210 to 230 degrees Celsius. Polycarbonate medical masterbatch requires higher temperatures including feed zone at 240 to 260 degrees Celsius, compression zone at 260 to 280 degrees Celsius, mixing zone at 270 to 290 degrees Celsius, and die zone at 270 to 290 degrees Celsius. Polyethylene medical masterbatch typically uses lower temperatures including feed zone at 150 to 170 degrees Celsius, compression zone at 170 to 190 degrees Celsius, mixing zone at 180 to 200 degrees Celsius, and die zone at 180 to 200 degrees Celsius.
Screw speed settings balance mixing effectiveness with gentle material handling. For medical color masterbatch with moderate colorant loading, typical screw speeds range from 80 to 150 rpm. Higher colorant loadings up to 20 percent may require higher speeds of 100 to 180 rpm to achieve adequate dispersion. UV stabilizer masterbatch with moderate loading typically uses speeds of 70 to 130 rpm. Antimicrobial masterbatch with lower additive loading often uses lower speeds of 60 to 120 rpm to maintain additive integrity. Optimal screw speed depends on additive sensitivity, carrier polymer viscosity, and required dispersion quality.
Feed rate settings must match throughput requirements while maintaining adequate residence time for mixing. For KTE Series counter rotating twin screw extruders, typical feed rates range from 50 to 1000 kg per hour depending on extruder size and formulation. Screw diameter of 40 mm typically handles 50 to 200 kg per hour. Screw diameter of 65 mm typically handles 150 to 500 kg per hour. Screw diameter of 90 mm typically handles 300 to 1000 kg per hour. Feed rate should be set to maintain appropriate residence time typically ranging from 30 to 90 seconds for medical applications depending on formulation complexity.
Vent settings depend on material characteristics and volatile content. For most medical masterbatch applications, first vent should be positioned after melting zone approximately at 60 to 70 percent of barrel length. Vent temperature should be set 20 to 30 degrees Celsius above material melt temperature to prevent condensation. Vent vacuum level typically ranges from atmospheric pressure to 500 mm Hg absolute depending on volatile content. Higher vacuum levels improve volatile removal but increase energy consumption and may cause material foaming in sensitive applications.
Die pressure settings ensure consistent strand formation and pellet quality. Strand die pressure typically ranges from 50 to 200 bar depending on formulation viscosity and throughput. Higher viscosities require higher die pressure for consistent strand formation. Die temperature should be set 5 to 10 degrees Celsius above final zone barrel temperature to ensure smooth flow through die. Die gap settings should produce strand diameter of 2 to 4 mm for typical medical masterbatch applications. Proper die settings ensure uniform strand diameter and surface quality.
Equipment Pricing
Equipment investment for counter rotating twin screw extruder systems varies based on size, configuration, and features required for specific medical masterbatch applications. Understanding pricing factors helps with budget planning and equipment selection.
KTE Series counter rotating twin screw extruder base machine pricing depends on screw diameter and length-to-diameter ratio. Models with 40 mm screw diameter and 40 L/D ratio typically range from USD 45,000 to USD 60,000. Models with 65 mm screw diameter and 40 L/D ratio typically range from USD 75,000 to USD 95,000. Models with 90 mm screw diameter and 40 L/D ratio typically range from USD 120,000 to USD 150,000. Larger models with 110 to 150 mm screw diameter range from USD 180,000 to USD 350,000 depending on specific configuration and features required.
Feeding systems add to total investment cost. Gravimetric feeding systems for multiple components typically cost USD 8,000 to USD 25,000 depending on number of feeders and capacity requirements. Single gravimetric feeder systems typically cost USD 5,000 to USD 12,000. Volumetric feeders provide lower-cost alternative at USD 3,000 to USD 8,000 but offer less accuracy for medical applications requiring precise formulation control. Feeding system selection should consider accuracy requirements and formulation complexity for specific medical masterbatch production needs.
Pelletizing system costs depend on pelletizing method and production capacity. Strand pelletizing systems typically cost USD 15,000 to USD 40,000 including water bath, strand guide, and pelletizer. Underwater pelletizing systems providing higher quality pellets typically cost USD 40,000 to USD 80,000 but require higher maintenance. Water ring pelletizing offers intermediate quality at cost of USD 25,000 to USD 50,000. Pelletizing system selection should balance quality requirements, production capacity, and operational complexity.
Control system and automation features affect total cost. Basic PLC control systems with touch screen interface typically cost USD 8,000 to USD 15,000 as part of standard equipment. Advanced control systems with data logging, batch tracking, and remote monitoring capabilities add USD 5,000 to USD 12,000. Sanitary design features for medical applications including enhanced cleanability, stainless steel construction, and CIP capabilities add USD 10,000 to USD 30,000 to equipment cost depending on extent of sanitary features required.
Complete system costs including extruder, feeding, pelletizing, and necessary ancillary equipment typically range from USD 80,000 to USD 300,000 for medical grade masterbatch production. Smaller complete systems for laboratory or pilot production typically range from USD 80,000 to USD 150,000. Medium production capacity systems typically range from USD 150,000 to USD 250,000. High-capacity production systems typically range from USD 250,000 to USD 500,000 depending on production requirements and configuration complexity. Additional costs for installation, training, and validation support typically add 10 to 20 percent to equipment costs.
Production Problems and Solutions
Medical grade masterbatch production may encounter various problems affecting product quality, yield, or processing efficiency. Understanding potential problems and implementing effective solutions ensures consistent production of high-quality medical masterbatch.
Color streaks and lines in finished masterbatch indicate inadequate dispersion of colorant pigments. This problem typically occurs when insufficient mixing energy fails to break pigment agglomerates properly. The root cause may include insufficient screw speed, inappropriate screw configuration, or excessive feed rate reducing residence time. Color streaks running in machine direction typically indicate inadequate distributive mixing along the flow path. Streaks in transverse direction often indicate poor initial mixing or material bridging in feed zone.
Solutions for color streaks include increasing screw speed by 10 to 30 percent to enhance mixing energy. Optimizing screw configuration with additional mixing elements improves distributive mixing. Reducing feed rate by 10 to 25 percent increases residence time for more complete mixing. Temperature profile adjustments improving material flow and reducing viscosity variations can help. For persistent streak problems, reformulation with better dispersant additives or finer pigment particle size may be necessary. Avoiding streak problems requires proper material preparation including adequate pigment dispersion before extrusion, appropriate screw configuration for specific pigment types, and regular equipment maintenance to ensure consistent mixing performance.
Bubble formation in masterbatch pellets indicates presence of moisture or volatiles. Bubbles can cause processing problems in downstream applications and affect final product quality. This problem typically occurs from inadequate material drying, insufficient venting during extrusion, or volatile components from additives. Moisture-related bubbles typically appear throughout pellets while volatile bubbles may concentrate near pellet centers. Bubble size and distribution provide clues to the specific cause.
Solutions for bubble problems start with thorough material drying before processing. Medical grade polymers should be dried to moisture content below 0.02 percent using appropriate drying conditions including temperature of 80 to 100 degrees Celsius for polypropylene, 70 to 90 degrees Celsius for polyethylene, and 100 to 120 degrees Celsius for polycarbonate for 2 to 4 hours. Vent system optimization including proper vent positioning and vacuum level improves volatile removal. Temperature profile adjustments reducing maximum temperature can minimize thermal degradation generating volatiles. Using additives with lower volatile content or pre-drying additives before use can reduce bubble formation. Avoiding bubble problems requires consistent material quality control, adequate drying procedures, and regular vent system maintenance ensuring proper operation.
Pellet color variation between batches indicates inconsistent processing conditions or material variations. This problem manifests as noticeable color differences between different production runs of the same formulation. Root causes may include material batch variations, parameter inconsistencies, or equipment wear affecting mixing performance. Color variation can cause problems in downstream applications where color consistency is critical, especially for medical packaging and medical device applications.
Solutions for color variation include implementing strict material quality control with incoming material testing and certificate of analysis verification. Documenting and standardizing processing parameters with tight tolerance ranges ensures batch-to-batch consistency. Regular calibration of temperature controllers, feeders, and other process equipment maintains parameter accuracy. Screw and barrel inspection identifies wear patterns affecting mixing performance. Implementing statistical process control monitors critical parameters and identifies trends before variations cause problems. Avoiding color variation requires comprehensive quality management system including material specifications, validated processes, and regular equipment maintenance ensuring consistent performance.
Material degradation causing discoloration or property changes indicates thermal stress during processing. Medical polymers may yellow, darken, or develop other discoloration from thermal degradation. Degraded material can affect downstream processing and final product performance. This problem typically occurs from excessive temperature, extended residence time, or shear heating. Discoloration severity provides indication of degradation extent with subtle changes indicating early degradation and severe changes indicating extensive degradation.
Solutions for material degradation include reducing processing temperatures by 10 to 30 degrees Celsius below current settings. Optimizing screw configuration to reduce shear and residence time decreases thermal stress. Reducing screw speed by 10 to 20 percent can reduce shear heating. Implementing vent systems removes degradation products and prevents further degradation. Using thermal stabilizers in formulation provides protection against thermal stress. Avoiding material degradation requires understanding thermal stability limits of specific polymers, implementing appropriate monitoring systems detecting early signs of degradation, and maintaining consistent processing conditions within established safe ranges.
Equipment contamination affecting masterbatch purity presents serious problem for medical applications. Contamination can come from wear debris, previous production materials, or external sources. Contaminated masterbatch can cause problems in medical applications where purity requirements are stringent. Contamination types include metallic particles from wear, color carryover from previous runs, or foreign material from processing environment.
Solutions for contamination problems include implementing comprehensive cleaning procedures between material changes using validated cleaning protocols. Using material handling systems designed for minimal contamination risk including closed conveying and storage systems. Implementing regular equipment inspection identifying wear before contamination occurs. Using wear-resistant materials and components extends service life and reduces debris generation. Establishing cleanroom or controlled environment for critical medical applications reduces external contamination risk. Avoiding contamination requires systematic approach including equipment design, cleaning procedures, material handling practices, and environmental control specifically addressing medical application requirements.
Inconsistent pellet size and shape affect downstream processing and material flow. This problem occurs when pelletizing system produces non-uniform pellets with size or shape variations. Root causes may include die wear, knife wear, or inconsistent strand formation. Pellet size variation can cause feeding problems in downstream extrusion processes and affect final product quality uniformity.
Solutions for pellet size inconsistency include regular die inspection and replacement when wear affects strand quality. Sharpening or replacing pelletizer knives ensures clean cutting. Optimizing water bath temperature and flow maintains consistent strand cooling preventing deformation. Adjusting feed rate and screw speed combination provides consistent strand formation. Implementing automated monitoring and feedback control of pelletizing parameters improves consistency. Avoiding pellet size problems requires regular maintenance schedule for pelletizing components, process optimization for consistent strand formation, and training operators to recognize and correct pellet quality problems promptly.
Maintenance and Care
Regular maintenance ensures consistent performance and extends equipment service life. Medical grade masterbatch production requires comprehensive maintenance program addressing equipment wear, cleanliness, and performance consistency.
Daily maintenance tasks should be performed by operators during normal production operations. Visual inspection should check for leaks, unusual vibrations, and abnormal sounds during operation. Temperature readings should be monitored and logged to ensure consistent thermal performance. Feeding systems should be checked for proper operation and accurate material delivery. Vent systems should be inspected for proper operation and filter condition. Pelletizing system should be monitored for consistent pellet quality. Daily maintenance logs should record inspection findings, parameter readings, and any corrective actions taken.
Weekly maintenance tasks require more detailed inspection and maintenance activities. Temperature control systems should be calibrated using reference thermometers to ensure accuracy. Feeder calibration should be verified using known weight samples confirming proper feeding accuracy. Die and knife inspection should check for wear affecting pellet quality. Barrel cooling systems should be checked for proper operation and water flow quality. Electrical systems should be inspected for loose connections, proper grounding, and adequate protection. Weekly maintenance should include cleaning of critical areas including feed zones, vent ports, and pelletizing components.
Monthly maintenance tasks address more comprehensive inspection and preventive maintenance. Screw and barrel inspection should examine wear patterns and assess remaining service life. Drive system inspection should check motor bearings, coupling condition, and drive belt or gearbox condition. Temperature sensors should be calibrated using reference standards ensuring accuracy within specifications. Pressure sensors and transmitters should be calibrated for accurate readings. Safety systems should be tested including emergency stops, overload protection, and safety interlocks. Monthly maintenance should include thorough cleaning of equipment interior surfaces accessible without major disassembly.
Semi-annual maintenance tasks involve more extensive maintenance activities requiring production downtime. Screw removal and inspection provides detailed wear assessment and enables cleaning not possible during routine operation. Barrel interior inspection checks for wear, scoring, and buildup affecting material flow and mixing. Drive system maintenance including motor bearing replacement, gearbox oil changes, and drive component inspection ensures reliable operation. Electrical system testing including thermal imaging, resistance measurement, and insulation testing identifies potential problems before failure. Heating elements should be inspected for continuity and proper operation. Semi-annual maintenance provides opportunity for thorough cleaning and validation of cleaning procedures.
Annual maintenance tasks represent comprehensive service requiring significant production planning. Complete screw and barrel inspection may reveal need for reconditioning or replacement based on wear assessment. Drive system overhaul including bearing replacement, gearbox service, and motor testing ensures long-term reliability. Control system review includes software updates, calibration verification, and data backup verification. Complete system cleaning provides opportunity for deep cleaning of areas not routinely accessible. Equipment performance review compares current performance to original specifications and establishes maintenance priorities for coming year. Annual maintenance supports validation requirements and ensures equipment continues meeting medical application requirements.
Preventive maintenance schedule should be documented and followed consistently. Documentation should include maintenance task descriptions, required intervals, responsible personnel, required tools and spare parts, acceptance criteria, and recording forms. Maintenance records should be retained for equipment lifecycle management and validation requirements. Predictive maintenance technologies including vibration analysis, thermal imaging, and oil analysis can predict failures before they occur reducing unplanned downtime. Spare parts inventory should be maintained based on maintenance history and supplier lead times ensuring parts availability when needed.
FAQ
Q: What are the key differences between counter rotating and co-rotating twin screw extruders for medical grade masterbatch production?
A: Counter rotating twin screw extruders provide positive displacement with relatively low shear rates making them suitable for shear-sensitive medical additives and colorants. Co-rotating extruders provide higher shear and better mixing but may be less suitable for sensitive materials. Counter rotating designs offer gentle material handling important for maintaining additive integrity. KTE Series counter rotating extruders are specifically optimized for medical applications requiring careful handling of sensitive components.
Q: What type of carrier polymer should be used for medical grade masterbatch?
A: Carrier polymer selection should match the end-use material and meet medical grade specifications. Common choices include medical grade polypropylene for polypropylene applications, medical grade polyethylene for polyethylene applications, and medical grade polycarbonate for polycarbonate applications. The carrier must have compatible melt flow characteristics and meet relevant medical standards including FDA, USP, and ISO requirements for the intended application.
Q: How often should medical grade masterbatch production equipment be cleaned?
A: Cleaning frequency depends on material changeover, production requirements, and validation status. For routine production with consistent material, daily cleaning of contact surfaces may be required. For material changes between different color masterbatches, complete cleaning procedures should be performed. Cleaning should always be performed before starting medical grade production after non-medical materials. Cleaning procedures should be validated and documented as part of quality management system for medical applications.
Q: What is the recommended moisture content for medical grade polymers before processing?
A: Most medical grade polymers require moisture content below 0.02 percent before processing. Specific requirements vary by polymer type with polycarbonate typically requiring 0.01 percent or lower due to sensitivity. Polypropylene and polyethylene typically require 0.02 to 0.05 percent moisture content. Proper drying using dehumidifying dryers at appropriate temperatures and times is essential for preventing bubble formation and material degradation during processing.
Q: How can I ensure consistent color in medical grade color masterbatch between batches?
A: Consistent color requires comprehensive quality control including material qualification with certificates of analysis, standardized processing parameters with tight tolerance ranges, regular equipment maintenance ensuring consistent performance, and color measurement with spectrophotometers for objective assessment. Implementing statistical process control monitors color variation and identifies trends before problems occur. Using standardized colorant sources and formulations reduces variation between batches.
Q: What safety considerations are important for medical grade masterbatch production?
A: Important safety considerations include proper ventilation for handling fine powders and dust control, appropriate personal protective equipment including respirators when handling powders, electrical safety for high-temperature equipment, mechanical safety for rotating components, and chemical safety for handling additives and colorants. Medical applications may require additional considerations including cleanroom protocols, contamination control measures, and validation documentation. Safety training should be comprehensive and regularly updated.
Q: How does screw configuration affect medical grade masterbatch quality?
A: Screw configuration determines mixing characteristics and shear stress experienced by materials. Medical grade masterbatch often requires configurations providing good mixing without excessive shear that could damage sensitive additives. Counter rotating designs typically use specialized mixing elements optimized for gentle yet effective dispersion. Configuration should be customized based on specific formulation requirements including additive types, loading levels, and dispersion quality requirements. KTE Series modular screw design allows configuration optimization for specific medical applications.
Q: What is the typical service life for screws and barrels in medical masterbatch production?
A: Service life depends on materials processed, operating conditions, and maintenance practices. Typical service life ranges from 3 to 5 years for standard construction screws and barrels in continuous operation. Hardened or coated components can provide extended service life up to 8 to 10 years. Medical grade materials with low abrasiveness typically provide longer service life compared to highly filled or abrasive compounds. Regular inspection and proper maintenance can extend service life and optimize cost per production hour.
Q: How can production yield be improved in medical grade masterbatch production?
A: Yield improvement starts with proper material handling and preparation minimizing losses. Optimizing processing parameters reduces off-specification material requiring disposal. Implementing scrap recycling systems recovers material from startup, shutdown, and process variations. Minimizing product transitions and changeovers reduces transition waste. Quality control systems catching problems early minimize waste generation. Regular maintenance prevents problems causing yield loss. Well-trained operators recognizing and correcting problems promptly improve overall yield.
Conclusion
Counter rotating twin screw extruders provide excellent capabilities for medical grade masterbatch production combining gentle material handling with effective mixing essential for sensitive medical additives and colorants. KTE Series counter rotating twin screw extruders from Nanjing Kerke Extruder Equipment Company incorporate specialized features specifically designed for medical applications including sanitary design, precise process control, and corrosion-resistant materials meeting the stringent requirements of healthcare industry applications.
Successful medical grade masterbatch production requires comprehensive approach addressing formulation development, process optimization, equipment selection, quality control, and maintenance. Understanding material characteristics, processing requirements, and regulatory standards enables development of formulations meeting medical application requirements. Proper parameter optimization including temperature profiles, screw speeds, feed rates, and venting ensures consistent production of high-quality masterbatch meeting medical specifications.
Quality management systems including material qualification, process validation, batch documentation, and regular testing ensure consistent production meeting medical industry requirements. Preventive maintenance programs ensure equipment reliability and performance consistency essential for medical applications where product quality and reliability are critical. Training programs ensure operators understand both technical requirements and quality considerations specific to medical applications.
Medical grade masterbatch production presents unique challenges due to stringent quality requirements, regulatory compliance needs, and zero tolerance for contamination. Counter rotating twin screw extruders provide the gentle mixing and precise control needed to meet these challenges while maintaining production efficiency. By selecting appropriate equipment, optimizing processes, implementing comprehensive quality systems, and maintaining equipment properly, medical masterbatch producers can reliably produce high-quality products meeting the demanding requirements of healthcare applications.