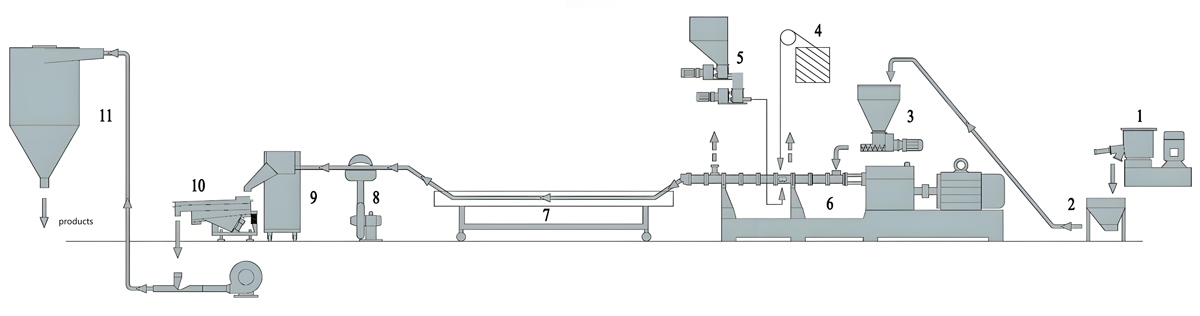
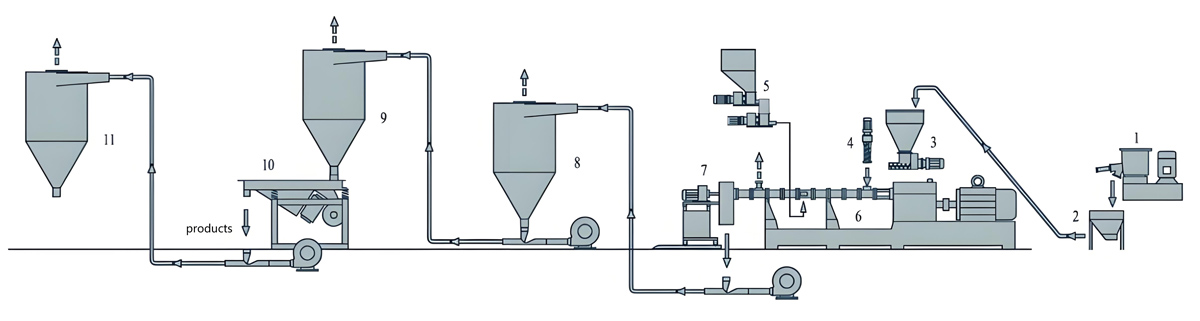
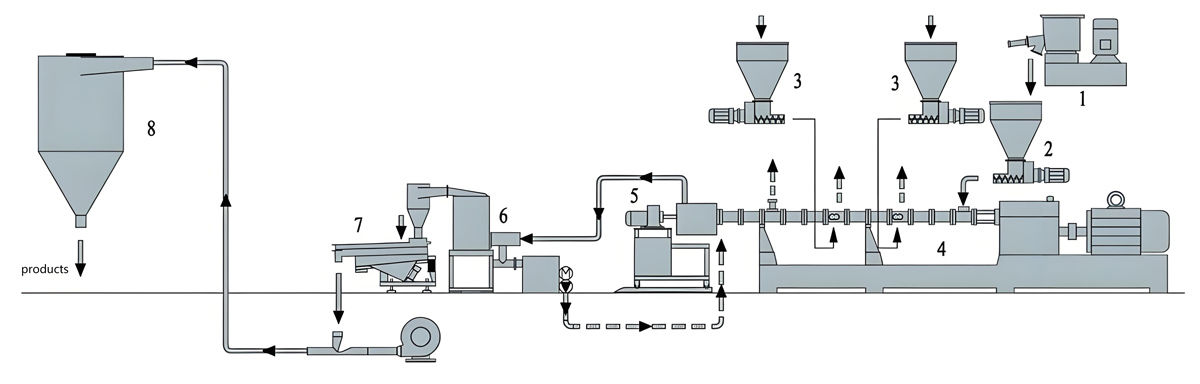
The food packaging industry demands masterbatch products that ensure safety, extend shelf life, and maintain product quality throughout the supply chain. Antibacterial masterbatch addresses these requirements by incorporating antimicrobial agents that inhibit bacterial growth on packaged food surfaces. The production of food contact antibacterial masterbatch demands precision equipment capable of maintaining strict quality standards while processing sensitive antimicrobial additives. Variable speed twin screw extruders have emerged as the preferred production technology for manufacturers seeking to deliver consistent, high-quality antibacterial masterbatch for food contact applications. The flexibility of variable speed operation enables optimization of processing conditions for each specific formulation, ensuring proper dispersion of antimicrobial agents while maintaining material integrity and regulatory compliance.
Introduction
Food safety concerns continue driving demand for antimicrobial packaging solutions that provide active protection against bacterial contamination. Antibacterial masterbatch enables plastic packaging manufacturers to incorporate antimicrobial functionality directly into their products, creating an additional safety barrier that complements traditional food safety practices. These specialized masterbatch products require precise manufacturing to ensure uniform dispersion of antimicrobial agents and maintain compliance with food contact regulations that vary by market.
The production of food contact antibacterial masterbatch presents unique technical challenges related to antimicrobial agent handling, regulatory compliance, and quality verification. Variable speed twin screw extrusion technology addresses these challenges through precise control over processing conditions that protect thermally sensitive antimicrobial compounds while achieving the mixing intensity required for proper dispersion. The flexibility to adjust screw speed throughout the production process enables optimization for different formulation compositions and production requirements.
This comprehensive guide addresses all aspects of food contact antibacterial masterbatch manufacturing using variable speed twin screw extrusion equipment. The content provides practical guidance for formulation development, process optimization, regulatory compliance, and quality assurance. By understanding these key factors, manufacturers can establish or improve their capabilities for producing high-quality antibacterial masterbatch that meets the demanding requirements of food contact applications.
Formulation Strategies for Food Contact Antibacterial Masterbatch
Regulatory-Compliant Antimicrobial Agents
Selection of appropriate antimicrobial agents represents the critical first step in food contact antibacterial masterbatch formulation. Regulatory requirements in target markets severely restrict which antimicrobial compounds may be used in food contact applications. In the United States, FDA regulations under 21 CFR Parts 170-199 govern substances permitted for food contact use. European Union Regulation 10/2011 establishes a positive list of authorized substances for plastic food contact materials. Similar regulatory frameworks exist in other major markets and must be consulted when developing formulations for international distribution.
Approved antimicrobial agents for food contact applications include silver-based compounds, zinc compounds, and certain organic antimicrobial agents. Silver ion-releasing agents provide broad-spectrum antimicrobial activity and have received regulatory approval in multiple jurisdictions. Nano-scale silver materials offer enhanced antimicrobial effectiveness but require additional safety documentation to address potential nanoparticle exposure concerns. Zinc pyrithione and related zinc compounds provide effective antimicrobial protection with well-established safety profiles for food contact use.
Carrier Resin Selection
Carrier resin selection for food contact antibacterial masterbatch must address both regulatory compliance and processing compatibility with antimicrobial agents. Polypropylene and polyethylene carriers offer excellent processing characteristics and broad regulatory acceptance for food contact applications. The specific grade selected must carry food contact compliance documentation from the resin supplier to support final masterbatch product regulatory claims.
Formulation compositions typically include 60-75% carrier resin to provide adequate carrier material for uniform antimicrobial distribution in end-use applications. Higher carrier loading reduces the concentration of antimicrobial agents in the final masterbatch but may simplify regulatory compliance by minimizing the effective concentration of potentially restricted compounds. Balance between antimicrobial effectiveness and regulatory compliance requires careful evaluation of intended application requirements and target market regulatory frameworks.
Synergistic Additive Systems
Effective antibacterial masterbatch formulations often incorporate synergistic additive systems that enhance antimicrobial performance beyond what individual components achieve alone. Antioxidant systems protect antimicrobial compounds from degradation during processing and storage, maintaining activity throughout the product shelf life. Stabilizers and UV absorbers provide similar protection against environmental degradation that could reduce antimicrobial effectiveness over time.
Carrier modification agents improve compatibility between antimicrobial compounds and the carrier resin matrix, promoting uniform dispersion and preventing migration of antimicrobial agents to the masterbatch surface during storage. Surface-treated antimicrobial compounds offer improved dispersion characteristics and reduced tendency toward agglomeration during processing. Processing aid packages including lubricants and flow modifiers ensure smooth extrusion processing while maintaining food contact compliance.
Production Process for Antibacterial Masterbatch
Antimicrobial Agent Preparation
Proper preparation of antimicrobial agents before incorporation into the extrusion formulation significantly impacts final product quality and performance. Powdered antimicrobial compounds require careful handling to prevent airborne exposure and ensure accurate weighing. Closed transfer systems and dust collection equipment protect operator safety while maintaining formulation accuracy. Pre-dispersion of antimicrobial agents in a small portion of carrier resin simplifies handling and improves distribution uniformity in the final formulation.
Moisture content of antimicrobial agents must be controlled to prevent degradation reactions during extrusion processing. Many antimicrobial compounds are sensitive to moisture and require drying prior to use. Silver-based antimicrobials may require special handling to prevent unwanted reduction reactions that could affect their activity. Documentation from antimicrobial suppliers regarding handling requirements and storage conditions should be reviewed and followed to ensure material integrity.
Compounding Process Optimization
Variable speed operation enables optimization of the compounding process throughout the production run based on real-time monitoring of processing conditions and product quality indicators. Initial processing at reduced screw speeds minimizes shear heating and thermal stress on antimicrobial compounds during the critical initial melting and mixing phases. As the process stabilizes and material temperature reaches steady-state conditions, screw speed may be increased to enhance mixing intensity and throughput capacity.
The extrusion process converts raw materials into homogeneous masterbatch through carefully controlled melting, mixing, and pumping operations. Material enters the extruder through the feed throat where screw geometry initiates melting and transport. The screw configuration in the melting zone determines how quickly material transitions from solid to molten state and establishes the foundation for subsequent mixing operations. Kneading blocks and mixing elements in the compounding zone subject material to intensive shearing that disperses antimicrobial agents throughout the carrier matrix.
Quality Control During Production
Production of food contact antibacterial masterbatch requires rigorous quality control to verify antimicrobial content, dispersion quality, and regulatory compliance. In-process sampling at regular intervals provides material for testing that verifies consistent production quality throughout the batch. Sampling points should include both the extrusion discharge and final pellet samples after cooling and classification to verify that quality is maintained through all processing stages.
Antimicrobial content verification through analytical testing confirms that target concentrations are achieved and maintained throughout production. Testing methods vary depending on the specific antimicrobial compound and may include atomic absorption spectroscopy for silver, inductively coupled plasma mass spectrometry for metals, or chromatographic methods for organic antimicrobials. Regular testing frequency depends on production volume and quality system requirements but typically includes samples from each production batch with more frequent testing for new formulations or following equipment adjustments.
Production Equipment for Antibacterial Masterbatch
KTE Series Variable Speed Twin Screw Extruder
The KTE Series variable speed twin screw extruder from Kerke provides the precise control and processing flexibility required for food contact antibacterial masterbatch production. Variable frequency drive systems enable smooth, continuous speed adjustment across the operating range without the step changes or mechanical stress associated with gear transmission changes. This speed flexibility proves essential for processing temperature-sensitive antimicrobial compounds while achieving the throughput rates required for commercial production.
The stainless steel construction of the KTE Series ensures food contact compliance throughout the production system. All material contact surfaces meet food grade surface finish requirements and are compatible with cleaning procedures necessary for food contact production. The modular barrel and screw element system enables optimization of screw configuration for specific antimicrobial formulations, providing the mixing intensity required for proper dispersion while minimizing unnecessary shear heating.
Advanced control systems in the KTE Series enable precise management of processing parameters throughout production. Individual zone temperature control maintains consistent thermal conditions despite varying material throughput or composition. Motor load monitoring provides real-time feedback on processing conditions that can indicate developing problems before they cause quality failures. Data logging capabilities support comprehensive quality documentation required for food contact regulatory compliance.
Material Handling Systems
Food contact production requires dedicated material handling systems that prevent contamination and maintain the purity required for regulatory compliance. Enclosed conveying systems protect materials from environmental contamination during transfer from storage to production equipment. Separate handling systems for antimicrobial agents prevent cross-contamination between different formulations and protect operators from exposure to concentrated antimicrobial compounds.
Gravimetric feeding systems provide superior formulation accuracy compared to volumetric alternatives, ensuring consistent antimicrobial concentration across production batches. Loss-in-weight feeders that continuously measure and adjust material flow based on actual weight provide the most accurate formulation control available. Multiple feeder configurations enable accurate addition of carrier resin, antimicrobial compounds, and additive systems through coordinated feeding sequences.
Downstream Processing Equipment
Pelletizing systems for food contact antibacterial masterbatch must maintain product purity and consistency while providing efficient production output. Underwater pelletizing systems offer advantages for food contact production including consistent pellet geometry, reduced contamination risk from external sources, and efficient cooling that minimizes thermal degradation. Strand pelletizing provides an alternative that may be preferred for lower volume production or formulations requiring specific pellet characteristics.
Pellet classification and packaging systems complete the production line with attention to contamination prevention and product protection. Metal detection systems verify absence of metallic contamination that could affect product safety or processing equipment. Packaging in food-grade containers with appropriate labeling provides traceability and regulatory compliance documentation throughout distribution. Automated packaging with inert atmosphere flushing extends product shelf life by displacing oxygen that could promote degradation of antimicrobial activity.
Parameter Settings for Food Contact Antibacterial Masterbatch
Temperature Profile Configuration
Temperature profile optimization for antibacterial masterbatch balances processing requirements with the thermal sensitivity of antimicrobial compounds. Most antimicrobial agents exhibit some degradation at elevated temperatures, requiring careful thermal management throughout the extrusion process. Initial temperature profiles should be established at the lower end of the acceptable processing range, with incremental increases only as necessary to achieve adequate processing conditions.
Typical barrel temperature profiles for antibacterial masterbatch range from 160-180 degrees Celsius in the feed zone to 200-230 degrees Celsius in the final mixing and pumping zones. The specific temperatures depend on carrier resin characteristics, antimicrobial compound thermal stability, and throughput requirements. Die temperatures should be maintained at or slightly below the final barrel zone temperature to prevent thermal degradation of material as it exits the extruder.
Screw Speed Optimization
Variable speed capability enables optimization of screw speed throughout the production process based on specific formulation requirements and processing conditions. Initial startup and initial melting phases typically benefit from reduced screw speeds of 150-250 rpm that minimize shear heating and provide extended time for complete melting. Higher speeds of 350-500 rpm may be appropriate during stable production phases when throughput optimization takes priority over minimizing shear exposure.
Formulations with highly temperature-sensitive antimicrobial compounds may benefit from extended processing at reduced speeds throughout the production run, accepting lower throughput in exchange for improved antimicrobial activity retention. Formulations with more thermally stable antimicrobials can utilize higher speeds during stable production phases, achieving better throughput efficiency while maintaining acceptable product quality.
Throughput and Residence Time Control
Throughput rate affects processing efficiency and product quality through impacts on residence time and mixing intensity. Higher throughput rates reduce residence time, which benefits thermally sensitive formulations but may reduce mixing intensity and compromise dispersion quality. Lower throughput rates provide extended mixing time that improves dispersion but increase exposure to thermal conditions that could degrade antimicrobial compounds.
Variable speed operation enables throughput optimization that balances these competing requirements for each specific formulation. Throughput rates for antibacterial masterbatch typically range from 150-400 kg/hour for production scale equipment, with the specific optimal rate depending on formulation composition, antimicrobial compound characteristics, and quality requirements. Regular monitoring of product quality indicators enables ongoing optimization of throughput parameters.
Equipment Price and Investment Considerations
Variable speed twin screw extrusion equipment for food contact antibacterial masterbatch requires investment levels appropriate to production scale and quality requirements. Laboratory and pilot scale systems suitable for product development and small batch production range from $45,000 to $110,000 depending on capacity and control system sophistication. Food contact production may require additional investment in enclosed material handling, metal detection, and documentation systems beyond standard laboratory equipment.
Production scale variable speed extruders with throughput capacity of 250-600 kg/hour typically range from $200,000 to $480,000 for complete systems including variable speed drives, food-grade material handling, and quality monitoring systems. Higher capacity systems exceeding 800 kg/hour require investments of $600,000 to $1,400,000 or more depending on configuration and automation level.
Food contact compliance introduces additional costs for validation documentation, regulatory compliance verification, and quality system maintenance that should be factored into investment analysis. The premium pricing available for food contact antibacterial masterbatch typically supports higher production margins that justify these additional investments for manufacturers targeting the food packaging market segment.
Production Problems, Causes, Solutions and Prevention
Antimicrobial Activity Loss During Processing
Problem: Produced masterbatch exhibits significantly reduced antimicrobial activity compared to expected levels based on formulation composition and analytical testing of raw materials. End-use testing fails to demonstrate the antimicrobial effectiveness required for intended applications. Customer complaints indicate inadequate performance in actual packaging applications.
Cause Analysis: Antimicrobial activity loss during processing typically results from thermal degradation of heat-sensitive antimicrobial compounds. Excessive barrel temperatures, extended residence time at elevated temperatures, or repeated thermal exposure during multiple processing passes can degrade antimicrobial compounds below effective concentration levels. Chemical reactions between antimicrobial agents and other formulation components may also reduce activity through sequestration or degradation reactions.
Solution: Reduce barrel temperatures systematically, beginning with the highest temperature zones, until analytical testing confirms acceptable antimicrobial retention. Review and optimize screw configuration to minimize residence time and reduce opportunities for thermal degradation. Implement single-pass processing protocols that eliminate unnecessary re-processing of material. Consult antimicrobial supplier technical support regarding specific handling requirements and compatible formulation components.
Prevention: Establish validated temperature profiles and processing parameters for each antimicrobial formulation before production. Implement real-time monitoring of processing conditions with automatic alerts when parameters deviate from validated ranges. Verify antimicrobial activity retention through regular testing of production samples. Maintain close communication with antimicrobial suppliers regarding formulation optimization and processing recommendations.
Non-Uniform Antimicrobial Dispersion
Problem: Produced masterbatch shows inconsistent antimicrobial performance across production batches or within individual batches. Some areas of packaged products demonstrate strong antimicrobial activity while others show no measurable effect. Testing reveals high variability in antimicrobial content across samples from the same production lot.
Cause Analysis: Non-uniform dispersion results from inadequate mixing intensity or improper handling of antimicrobial compounds before or during extrusion. Agglomeration of antimicrobial particles prevents uniform distribution throughout the carrier matrix. Feeding problems that introduce antimicrobial material in pulses rather than continuous flow cause localized concentration variations. Improper screw configuration that does not provide sufficient mixing intensity fails to break apart agglomerates and distribute antimicrobial agents uniformly.
Solution: Modify screw configuration to increase mixing intensity through additional kneading blocks or specialized mixing elements. Implement pre-dispersion of antimicrobial agents to reduce particle size before addition to extrusion formulation. Verify feeder calibration and operation to ensure continuous, uniform addition throughout production. Consider switch to more readily dispersible antimicrobial forms or surface-treated materials that resist agglomeration.
Prevention: Validate mixing efficiency through dispersion testing before production qualification. Maintain formulation consistency by using standardized antimicrobial preparation procedures. Implement statistical process control monitoring that detects dispersion variability early in production. Establish incoming quality specifications for antimicrobial compounds that ensure consistent particle size and dispersion characteristics.
Regulatory Compliance Documentation Gaps
Problem: Quality audits or customer inquiries reveal missing or inadequate documentation supporting food contact regulatory compliance. Raw material compliance documentation is incomplete or outdated. Production records do not provide sufficient traceability to demonstrate compliance with regulatory requirements.
Cause Analysis: Documentation gaps typically result from inadequate quality system procedures for maintaining food contact compliance documentation. Supplier changes that introduce new sources of compliant materials without updating documentation systems. Insufficient understanding of specific regulatory requirements for target markets leads to incomplete documentation efforts. Production record systems that do not capture information necessary for regulatory traceability.
Solution: Conduct comprehensive audit of existing documentation against regulatory requirements for target markets. Obtain required compliance documentation from raw material suppliers, updating all outdated or incomplete materials. Implement documentation control procedures that ensure compliance documentation remains current and accessible. Develop production record formats that capture all information necessary for regulatory traceability and customer inquiries.
Prevention: Establish clear responsibility for food contact compliance documentation within the quality organization. Implement supplier qualification procedures that require complete compliance documentation as part of approval. Establish regular review schedules that verify documentation currency and completeness. Train production and quality personnel on documentation requirements and procedures.
Cross-Contamination Between Formulations
Problem: Antibacterial masterbatch production shows evidence of contamination from previously produced formulations. Analytical testing reveals presence of unauthorized antimicrobial compounds or other materials that should not appear in current production. Customer complaints indicate unexpected characteristics in delivered products.
Cause Analysis: Cross-contamination occurs when material from previous production runs remains in the extruder or handling systems and contaminates subsequent production. Inadequate purging between formulation changes leaves residual material that appears in subsequent batches. Material handling systems without proper segregation allow cross-contamination during transfer and storage. Equipment maintenance activities introduce contamination from lubricants, cleaning materials, or repair components.
Solution: Implement comprehensive cleaning and purging procedures between production runs of different formulations. Extend purge material quantities and purging time to ensure complete removal of previous materials. Clean material handling systems including feeders, hoppers, and conveyors before formulation changes. Verify cleaning effectiveness through analytical testing before releasing production for shipment.
Prevention: Establish dedicated equipment or formulation scheduling that minimizes changeover frequency. Implement validated cleaning procedures with verification testing for each equipment configuration. Maintain production scheduling records that enable identification of potential contamination sources. Use physical segregation or dedicated equipment for formulations with significant contamination sensitivity.
Pellet Quality Issues Affecting Downstream Processing
Problem: Produced masterbatch pellets exhibit quality issues that cause problems during downstream processing by customers. Issues include inconsistent pellet size, excessive fines, surface defects, or handling difficulties that affect customer production efficiency and product quality.
Cause Analysis: Pellet quality problems originate from issues in the extrusion process, pelletizing operation, or cooling system that affect final pellet characteristics. Inconsistent extrusion flow causes varying strand diameter that translates to variable pellet size. Improper cooling causes thermal stress that produces surface defects or pellet breakage during handling. Die plate problems including worn holes, misalignment, or material buildup affect strand quality and subsequent pellet characteristics.
Solution: Verify and optimize extrusion parameters including pressure stability, temperature consistency, and throughput uniformity. Inspect and clean die plate to remove buildup and restore proper hole geometry. Adjust cooling system settings to achieve appropriate cooling rate without thermal shock that causes stress defects. Implement pellet classification and screening to remove fines and oversized particles before packaging.
Prevention: Establish regular die plate inspection and cleaning schedules based on production volume and material characteristics. Implement continuous monitoring of pellet quality indicators including size distribution and fines content. Maintain pelletizing equipment including knife blades and cooling systems to manufacturer specifications. Develop customer feedback integration that identifies pellet quality issues before they affect significant production volumes.
Maintenance and Care
Food Contact Compliance Maintenance Requirements
Maintenance activities for food contact production equipment require attention to both mechanical reliability and contamination prevention. Lubricants and maintenance materials must be verified as food-grade and suitable for food contact applications before use on production equipment. Maintenance procedures should minimize opportunities for maintenance materials to contact product-contact surfaces. Equipment design should facilitate cleaning and inspection while preventing contamination from maintenance activities.
Scheduled maintenance should include verification of food contact surface condition and cleaning system effectiveness. Wear patterns on screws, barrels, and material handling components should be monitored to identify conditions that could affect product purity or quality. Replacement parts should be verified as food-grade or from approved suppliers meeting food contact requirements.
Equipment Cleaning Procedures
Effective cleaning procedures ensure that food contact equipment maintains the purity required for regulatory compliance and product quality. Cleaning validation should verify removal of previous formulation materials and absence of cleaning residue that could contaminate subsequent production. Cleaning procedures should address all product contact surfaces including barrel, screw elements, die plate, pelletizing system, and material handling equipment.
Purging compounds specifically designed for food contact applications enable efficient cleaning between formulation changes while meeting food safety requirements. Material compatibility with purging compounds should be verified before adoption in production procedures. Validation testing should confirm cleaning effectiveness through analytical methods appropriate for detecting formulation residue at required sensitivity levels.
Preventive Maintenance Scheduling
Preventive maintenance scheduling should balance equipment reliability requirements with production efficiency and food contact compliance needs. Component wear rates depend on production volume, formulation abrasiveness, and operating conditions and should be monitored to optimize maintenance timing. Critical wear components including screw elements, bearings, and seals should be inspected and replaced according to documented schedules based on actual wear observations.
Documentation of maintenance activities supports food contact compliance by demonstrating that equipment has been properly maintained throughout its operating history. Maintenance records should capture all activities including routine servicing, component replacements, and equipment adjustments. Integration of maintenance records with production records enables complete traceability for regulatory and customer quality inquiries.
FAQ
What regulatory requirements apply to food contact antibacterial masterbatch?
Food contact antibacterial masterbatch must comply with applicable regulations in each target market where products will be sold. In the United States, FDA regulations under 21 CFR Parts 170-199 establish requirements for substances used in food contact materials. The European Union requires compliance with Regulation 10/2011 and its subsequent amendments regarding authorized substances for plastic food contact materials. Similar regulatory frameworks exist in other major markets including China, Japan, and many other countries. Manufacturers must verify compliance for each specific market and maintain documentation supporting compliance claims. Migration testing may be required to demonstrate that antimicrobial agents do not migrate into food at levels exceeding regulatory limits.
How is antimicrobial effectiveness verified for food contact masterbatch products?
Antimicrobial effectiveness testing evaluates the ability of treated packaging to inhibit bacterial growth under standardized test conditions. Common test methods include ISO 22196 for measuring antibacterial activity on plastic surfaces and JIS Z 2801 for antimicrobial treated products. Testing evaluates reduction in bacterial populations after specified contact times compared to untreated control materials. Testing should be conducted with bacterial species relevant to intended food applications and should verify effectiveness throughout the expected product shelf life.
What shelf life can be expected for food contact antibacterial masterbatch?
Shelf life for food contact antibacterial masterbatch depends on formulation composition, storage conditions, and packaging protection. Properly formulated and stored masterbatch typically maintains acceptable antimicrobial activity for 12-24 months. Antimicrobial activity may degrade over time due to chemical reactions, migration from the carrier matrix, or interaction with environmental factors. Storage in cool, dry conditions with protection from light helps preserve antimicrobial effectiveness. Packaging with barrier properties prevents moisture and oxygen exposure that could accelerate degradation.
Can antibacterial masterbatch be used with recycled polymer carriers?
Antibacterial masterbatch formulations can incorporate recycled polymer carriers subject to regulatory compliance verification and quality consistency requirements. Post-consumer recycled materials introduce variability and potential contamination concerns that require careful evaluation for food contact applications. Regulatory frameworks vary regarding recycled content in food contact materials, with some jurisdictions requiring virgin materials for certain applications. Technical compatibility between recycled carriers and antimicrobial compounds should be verified through testing before commercial production.
What application concentrations are typically used for antibacterial masterbatch?
Application concentrations for antibacterial masterbatch depend on specific antimicrobial agents, target bacterial species, and application requirements. Typical addition rates range from 1-5% by weight in final plastic articles, with higher concentrations used for applications requiring more aggressive antimicrobial protection. The specific effective concentration depends on antimicrobial agent potency, carrier resin compatibility, and processing conditions that affect final antimicrobial availability. Application concentration should be optimized through testing with actual production formulations and representative bacterial challenge conditions.
Conclusion
Food contact antibacterial masterbatch production requires careful attention to formulation development, processing optimization, regulatory compliance, and quality assurance throughout the manufacturing operation. Variable speed twin screw extrusion technology provides the precise control and processing flexibility necessary to address the unique challenges of antibacterial masterbatch production while maintaining the throughput efficiency required for commercial viability.
The KTE Series variable speed twin screw extruder from Kerke delivers the capabilities required for food contact antibacterial masterbatch manufacturing, including precise temperature control, variable speed flexibility, and food-grade construction throughout the production system. Combined with appropriate material handling, quality monitoring, and documentation systems, these extruders enable manufacturers to produce high-quality antibacterial masterbatch that meets demanding food contact regulatory requirements.
The growing demand for antimicrobial packaging solutions driven by food safety concerns and extended shelf life requirements creates significant market opportunities for manufacturers with established capabilities in food contact antibacterial masterbatch production. Investment in appropriate production technology, quality systems, and regulatory compliance expertise positions manufacturers to capture these opportunities while building sustainable competitive advantages in this specialized market segment.